Unlocking Catalytic Performance: The Critical Role of Brønsted to Lewis Acid Site Ratio in H-ZSM-5 Zeolite Design
This article provides a comprehensive analysis of the Brønsted to Lewis (B/L) acid site ratio in H-ZSM-5 zeolites and its decisive influence on catalytic behavior.
Unlocking Catalytic Performance: The Critical Role of Brønsted to Lewis Acid Site Ratio in H-ZSM-5 Zeolite Design
Abstract
This article provides a comprehensive analysis of the Brønsted to Lewis (B/L) acid site ratio in H-ZSM-5 zeolites and its decisive influence on catalytic behavior. Targeting researchers and professionals in catalysis and chemical engineering, the review explores the foundational definitions and synthesis-dependent origins of these acid sites. It details advanced methodologies for the precise characterization, quantification, and intentional modulation of the B/L ratio. The discussion extends to troubleshooting common challenges in achieving target ratios and optimizing catalyst performance for specific reactions like methanol-to-hydrocarbons (MTH) and biomass conversion. Finally, the article compares the performance of catalysts with tailored B/L ratios against conventional benchmarks, validating strategies for achieving superior selectivity, activity, and stability. This synthesis offers a strategic guide for designing next-generation zeolite catalysts with application-specific acid site architectures.
Acid Site Fundamentals: Decoding Brønsted vs. Lewis Chemistry in H-ZSM-5 Zeolites
Zeolites are microporous, crystalline aluminosilicates that serve as pivotal solid acid catalysts in the chemical and petrochemical industries. Their catalytic activity is primarily governed by the nature, strength, and density of their acid sites. In the context of ongoing research on the Brønsted to Lewis (B/L) acid site ratio in H-ZSM-5, a thorough understanding of these sites' definitions and characteristics is fundamental.
Fundamental Definitions
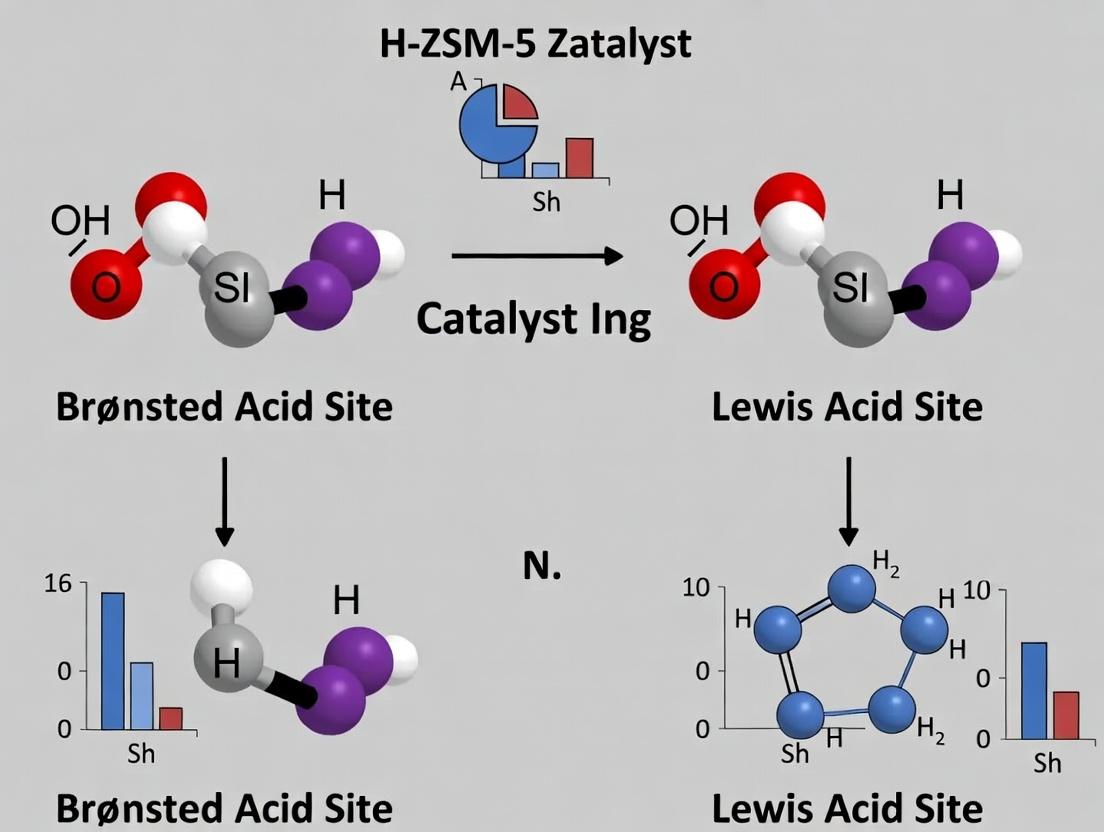
Brønsted Acid Sites (BAS) in zeolites are proton donors, originating from the bridging hydroxyl groups (Si–OH–Al) associated with framework aluminum atoms. The incorporation of a trivalent Al³⁺ ion into the tetrahedral SiO₄ framework creates a negatively charged site, which is balanced by a proton. This proton confers strong Brønsted acidity, enabling reactions like cracking, isomerization, and alkylation.
Lewis Acid Sites (LAS) are electron pair acceptors. In zeolites, they arise from:
- Extra-framework aluminum (EFAI): Aluminum species ejected from the framework during thermal or hydrothermal treatment (e.g., calcination, steaming).
- Coordinated unsaturated sites: Such as tri-coordinated framework aluminum or cationic sites (e.g., Zn²⁺, Cu²⁺ introduced via ion exchange).
The interplay between BAS and LAS, quantified as the B/L ratio, critically influences catalyst activity, selectivity, and deactivation in H-ZSM-5, particularly in reactions like methanol-to-hydrocarbons (MTH) and fluid catalytic cracking (FCC).
Quantitative Characterization Data
The concentration and ratio of acid sites are typically measured using a combination of probe molecule adsorption followed by spectroscopic or temperature-programmed desorption techniques.
Table 1: Common Quantitative Techniques for Acid Site Characterization in H-ZSM-5
| Technique | Probe Molecule | Target Site | Measurable Parameters | Typical Data for H-ZSM-5 (Si/Al=15) |
|---|---|---|---|---|
| NH₃-TPD | Ammonia (NH₃) | BAS & LAS (Total Acidity) | Acid site concentration (μmol/g), strength distribution | Total acidity: ~0.8-1.2 mmol NH₃/g |
| Pyridine FTIR | Pyridine (C₅H₅N) | BAS & LAS (Differentiated) | BAS concentration (B, μmol/g), LAS concentration (L, μmol/g), B/L Ratio | B: ~0.35 mmol/g, L: ~0.15 mmol/g, B/L: ~2.3 |
| 2,6-Di-tert-butylpyridine FTIR | 2,6-DTBPy | BAS (Sterically hindered) | Accessibility of strong BAS | Accessible BAS: ~60-80% of total BAS |
| IPy FTIR | Isobutylamine (i-C₄H₉NH₂) | BAS Strength | Number of strong BAS | Varies with treatment; steaming reduces strong BAS. |
Table 2: Effect of Post-Synthetic Treatment on B/L Ratio in H-ZSM-5 (Representative data from recent studies)
| Treatment Condition | Effect on Framework Al | Effect on EFAI | Typical B/L Ratio Change | Primary Consequence |
|---|---|---|---|---|
| Mild Calcination | Preserved | Minimal creation | High (>5) | High BAS activity, prone to coking. |
| Severe Steaming | Extensive dealumination | High generation | Low (<1) | Increased LAS, enhanced hydride transfer, reduced activity. |
| Acid Leaching | Removes some EFAL | Removes EFAL | Increases vs. steamed sample | Can tailor B/L by selective Al removal. |
| Metal Incorporation | Preserved | Introduces cationic LAS | Decreases significantly | Creates multifunctional (acid+redox) sites. |
Experimental Protocols for Key Characterization
Protocol 1: Temperature-Programmed Desorption of Ammonia (NH₃-TPD)
- Pretreatment: Load 100 mg of H-ZSM-5 pellet (250-425 μm) into a quartz U-tube reactor. Heat to 500°C (10°C/min) under He flow (30 mL/min) for 1 hour to clean the surface.
- Ammonia Saturation: Cool to 100°C. Switch to a 5% NH₃/He gas mixture (30 mL/min) for 30 minutes.
- Physisorbed NH₃ Removal: Flush with He at 100°C for 1-2 hours to remove weakly bound/physisorbed ammonia.
- Desorption: Heat the sample from 100°C to 700°C at a rate of 10°C/min under He flow (30 mL/min).
- Detection: Monitor desorbed NH₃ using a thermal conductivity detector (TCD) or mass spectrometer (MS, m/z=16). Quantify total acidity by integrating the TCD signal and calibrating with known NH₃ pulses.
Protocol 2: Pyridine Adsorption Fourier-Transform Infrared Spectroscopy (Py-FTIR)
- Wafer Preparation: Press 10-15 mg of zeolite powder into a self-supporting wafer. Place it in a controlled-environment IR cell with CaF₂ windows.
- In Situ Pretreatment: Evacuate the cell (<10⁻³ Pa) and heat the wafer to 450°C for 2 hours to remove adsorbed water and contaminants.
- Pyridine Adsorption: Cool to 150°C. Expose the wafer to pyridine vapor (equilibrium pressure ~0.5 Torr) for 15 minutes.
- Evacuation: Evacuate at 150°C for 30 minutes to remove physisorbed pyridine.
- Spectra Acquisition: Record the IR spectrum in the 1400-1700 cm⁻¹ region at 150°C.
- Quantification: Use the molar extinction coefficients to calculate site concentrations:
- BAS: Band at ~1545 cm⁻¹ (PyH⁺).
[B] (μmol/g) = (IA * Aw) / (ε * w), where IA is integrated absorbance, Aw wafer area (cm²), ε extinction coefficient (1.67 cm/μmol for 1545 cm⁻¹), and w wafer mass (g). - LAS: Band at ~1455 cm⁻¹ (coordinated pyridine). Use ε = 2.22 cm/μmol.
- BAS: Band at ~1545 cm⁻¹ (PyH⁺).
Visualization of Concepts and Workflows
Formation of BAS and LAS in H-ZSM-5
Py-FTIR Acid Site Characterization Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Materials for Acid Site Characterization
| Item | Function / Role in Research | Key Notes for H-ZSM-5 Studies |
|---|---|---|
| H-ZSM-5 Zeolite (NH₄⁺ or H⁺ form) | The core catalyst material under investigation. | Controlled Si/Al ratio (e.g., 15, 25, 40) is crucial. Particle size and binder content affect diffusion. |
| Anhydrous Ammonia (5% in He) | Probe molecule for NH₃-TPD to measure total acidity and strength. | Must be high-purity, dry. Can be generated in-situ from ammonium salts. |
| Anhydrous Pyridine | Selective IR probe to distinguish and quantify BAS vs. LAS. | Must be thoroughly dried over molecular sieves. Toxic; handle in fume hood. |
| 2,6-Di-tert-butylpyridine (2,6-DTBPy) | Bulky IR probe to assess external/accessible BAS only. | Steric hindrance prevents diffusion into micropores, probing only surface sites. |
| Isobutylamine | Probe for temperature-programmed surface reaction (TPSR) to count strong BAS. | Decomposes to NH₃ and isobutene only on strong acid sites. |
| High-Purity Carrier Gases (He, Ar, N₂) | For pretreatment, adsorption, and desorption steps. | Essential to use gases with oxygen/water traps (<1 ppm H₂O/O₂) to prevent sample alteration. |
| KBr or CaF₂ Windows/ Powder | For FTIR sample cells (windows) or preparing transmission pellets (KBr). | CaF₂ windows allow measurements down to ~1200 cm⁻¹; KBr is for diffuse reflectance. |
| Calibration Gas (e.g., 1% NH₃ in He) | For quantitative calibration of TCD response in NH₃-TPD. | Required to convert desorption peak area to micromoles of acid sites per gram. |
Within the broader research on Brønsted to Lewis acid site (B/L) ratio comparison in H-ZSM-5, understanding the genesis of these sites is paramount. This whitepaper details how synthesis conditions and post-synthetic modifications dictate the concentration, strength, and ratio of Brønsted (B) and Lewis (L) acid sites in H-ZSM-5 zeolites. This ratio critically influences catalytic performance in hydrocarbon conversion, methanol-to-olefins, and other key industrial processes.
Synthesis of H-ZSM-5: Creating the Brønsted Acid Framework
The primary source of Brønsted acidity in H-ZSM-5 is the bridging hydroxyl group (Si–OH–Al) formed when a tetrahedral aluminum atom is incorporated into the siliceous MFI framework. The synthesis directly controls the initial number of these sites.
Key Synthesis Parameters & Quantitative Data
The following parameters govern Al incorporation and initial B site density.
Table 1: Synthesis Parameters Governing B Site Formation in H-ZSM-5
| Parameter | Typical Range | Effect on B Sites (Si/Al Ratio) | Key Mechanism |
|---|---|---|---|
| SiO₂/Al₂O₃ in Gel | 30 to ∞ | Directly sets theoretical upper limit of Al content. Lower ratio = higher B site density. | Determines available Al for framework incorporation. |
| Structure-Directing Agent (SDA) | e.g., TPAOH, TBAOH | Influences crystallization rate, phase purity, and Al distribution. | Can affect Al zoning (homogeneous vs. gradient distribution). |
| Alkali Metal (Na⁺) Content | Minimized (<0.05 wt%) | High Na⁺ leads to Na-ZSM-5; requires ion exchange for H⁺. Residual Na⁺ neutralizes B sites. | Competition between Na⁺ and SDA for Al charge balancing. |
| Crystallization Temp & Time | 150-180°C, 24-72 hrs | Under-crystallization yields extra-framework Al (EFAL, L sites). Over-crystallization can dealuminate. | Completeness of Al integration into the framework. |
| Aging & Stirring | Variable | Affects gel homogeneity and nucleation, influencing uniform Al distribution. | Impacts reproducibility of acid site distribution. |
Experimental Protocol: Hydrothermal Synthesis of H-ZSM-5
Protocol 1: Standard Hydrothermal Synthesis for Controlled Si/Al Ratio
- Solution A: Dissolve sodium aluminate (NaAlO₂) in deionized water.
- Solution B: Dilute tetrapropylammonium hydroxide (TPAOH, 40% aqueous) in water, then add colloidal silica (e.g., Ludox HS-40) under vigorous stirring.
- Slowly add Solution A to Solution B. Stir the resultant gel for 2-4 hours at room temperature until homogeneous.
- Transfer the gel to a Teflon-lined stainless steel autoclave.
- Crystallize: Heat in a convection oven at 170°C for 48 hours under autogenous pressure.
- Quench & Recover: Cool rapidly, filter, and wash the solid product with deionized water until filtrate pH ~7-8.
- Dry: Dry at 100°C overnight.
- Calcination: Heat in static air to 550°C at 1°C/min, hold for 6 hours to remove the organic SDA (TPA⁺).
- Ion Exchange: Convert Na-ZSM-5 to NH₄-ZSM-5 by stirring in 1M NH₄NO₃ solution (10 mL/g zeolite) at 80°C for 2 hours (repeat 3x). Wash, dry.
- Final Activation: Calcine NH₄-ZSM-5 at 450-500°C (2°C/min, hold 4h) to produce H-ZSM-5 (NH₃ evolution creates H⁺).
Post-Synthetic Treatments: Modifying B/L Ratio
Treatments after synthesis are the primary tools for tuning the B/L ratio by selectively creating or destroying sites.
Dealumination: Converting B Sites to L Sites
Controlled extraction of framework Al creates silanol nests and extra-framework aluminum (EFAL) species, which are Lewis acidic.
Methods:
- Steaming: High-temperature treatment with steam hydrolyzes Si–O–Al bonds, ejecting Al as EFAL.
- Acid Leaching (e.g., HCl, HNO₃): Can remove some EFAL, leaving behind a more siliceous framework with strong B sites and reduced L sites. Complex effects on B/L ratio.
Desilication: Enhancing Mesoporosity & Influencing Acidity
Alkaline treatment (e.g., NaOH) selectively extracts silicon, creating mesopores. It can also redistribute Al, affecting acidity.
Table 2: Post-Synthetic Treatments and Their Impact on B/L Ratio
| Treatment | Typical Conditions | Primary Effect on Sites | Resulting B/L Ratio Trend | Secondary Effect |
|---|---|---|---|---|
| Steaming (Mild) | 500-600°C, 100% H₂O, 1-5h | Partial dealumination. Creates EFAL (L sites). | Decreases | Increases strength of remaining B sites. |
| Steaming (Severe) | >700°C, H₂O, prolonged | Extensive dealumination. Destroys B sites, creates abundant EFAL. | Greatly Decreases | Can cause structural degradation. |
| Acid Leaching (Mild) | e.g., 0.1M HNO₃, 80°C, 2h | Selectively removes non-framework Al (some L sites). | Increases | Can heal silanol nests, slightly reducing B site count. |
| Desilication | 0.1-0.5M NaOH, 65°C, 30min | Creates mesopores; can realuminate framework or create EFAL. | Variable | Depends on Al zoning; often increases accessibility to all sites. |
Experimental Protocol: Post-Synthetic Steaming and Acid Leaching
Protocol 2: Sequential Steaming and Acid Leaching to Tune B/L Ratio
- Starting Material: Begin with parent H-ZSM-5 (Si/Al = 40).
- Steaming (to create L sites):
- Place 2g of H-ZSM-5 in a quartz boat inside a tubular furnace.
- Pre-heat furnace to desired temperature (e.g., 550°C) under dry N₂ flow (50 mL/min).
- Switch N₂ flow through a water saturator at 80°C to introduce steam.
- Maintain sample under steam flow for 3 hours.
- Cool to room temperature under dry N₂.
- Acid Leaching (to remove some L sites):
- Stir 2g of the steamed zeolite in 100 mL of 0.1M HNO₃ solution.
- Heat at 80°C for 2 hours with reflux.
- Filter, wash thoroughly with deionized water, and dry at 100°C overnight.
- Re-activation: Calcine the leached sample at 450°C for 2 hours in dry air to produce the final modified H-ZSM-5.
Characterization Data: Quantifying B and L Sites
The B/L ratio is typically quantified using probe molecule adsorption followed by spectroscopic or calorimetric techniques.
Table 3: Characterization Methods for Acid Site Quantification
| Method | Probe Molecule | Information Obtained | Typical Data for H-ZSM-5 (Si/Al=40) |
|---|---|---|---|
| NH₃-TPD | Ammonia (NH₃) | Total acid site density & strength distribution. | Total acidity: ~0.4-0.6 mmol NH₃/g. Cannot reliably distinguish B/L. |
| Pyridine FTIR | Pyridine | Quantitative B and L site concentration. | Parent: B ~0.35 mmol/g, L ~0.05 mmol/g (B/L ~7). Steamed: B ~0.20 mmol/g, L ~0.15 mmol/g (B/L ~1.3). |
| i-Propylamine TPD | Isopropylamine | Counts Brønsted sites exclusively (decomposes to propene + NH₃). | Parent H-ZSM-5: ~0.32 mmol/g. Correlates with framework Al. |
The Scientist's Toolkit: Key Research Reagent Solutions
Table 4: Essential Materials for H-ZSM-5 Acidity Research
| Reagent/Material | Function in Research | Key Consideration |
|---|---|---|
| Tetrapropylammonium Hydroxide (TPAOH) | Structure-directing agent (SDA) for MFI synthesis. | High purity (>40% aq.) is critical. Determines crystal size/morphology. |
| Colloidal Silica (Ludox HS-40) | High-purity silica source for synthesis. | Provides reactive Si for framework building. Consistent particle size is key. |
| Sodium Aluminate (NaAlO₂) | Aluminum source for framework incorporation. | Must be fresh to avoid hydrolysis/condensation. Controls Si/Al ratio. |
| Ammonium Nitrate (NH₄NO₃) | For ion exchange to convert Na-ZSM-5 to its H-form. | Multiple exchanges at elevated temperature ensure complete Na⁺ removal. |
| Pyridine (spectroscopic grade) | Probe molecule for FTIR differentiation of B and L sites. | Must be thoroughly dried and purified. Adsorption is done under vacuum. |
| Nitric Acid (HNO₃, high purity) | For post-synthetic acid leaching treatments. | Mineral acid preferred over HCl to avoid introducing chloride impurities. |
Visualized Pathways and Workflows
Title: H-ZSM-5 Synthesis and Post-Synthetic Modification Workflow
Title: Mechanism of Dealumination Creating Lewis Sites
Within a broader thesis investigating Brønsted to Lewis acid site ratio comparisons in H-ZSM-5, understanding the fundamental electronic structure of acid sites is paramount. This whitepaper delves into the quantum chemical and spectroscopic foundations that govern the strength and behavior of both Brønsted and Lewis acid sites in H-ZSM-5 zeolites. The interplay between these sites dictates catalytic performance in key reactions relevant to petrochemical refining and, by methodological analogy, to drug development processes such as API synthesis and catalysis.
Electronic Structure of Acid Sites
Brønsted Acid Sites (BAS)
The primary Brønsted acid site in H-ZSM-5 is a bridging hydroxyl group (Si-OH-Al). Its acidity arises from the polarizability of the O-H bond, influenced by the surrounding aluminosilicate framework.
Key Electronic Features:
- Local Structure: The proton is covalently bonded to a framework oxygen, which bridges a tetrahedral silicon and a tetrahedral aluminum atom.
- Acidity Origin: The electron-withdrawing effect of the trivalent aluminum adjacent to the bridging oxygen weakens the O-H bond, facilitating proton donation.
- Probe Molecules: The stretching frequency (ν(OH)) of the bridging hydroxyl, typically around 3605-3610 cm⁻¹ in IR spectroscopy, serves as an indirect measure of acidity strength. A lower frequency indicates a stronger acid site due to greater O-H bond weakening.
Lewis Acid Sites (LAS)
Lewis acid sites in H-ZSM-5 are commonly associated with extra-framework aluminum (EFAl) species or framework defects. They function as electron pair acceptors.
Key Electronic Features:
- Composition: Typically trigonal or tetrahedral coordinatively unsaturated Al³⁺ ions, not fully integrated into the silicate framework.
- Acidity Origin: The electron deficiency on the Al center allows it to accept electron density from adsorbates.
- Characterization: Identified via adsorption of probe molecules like pyridine (Py-IR), showing characteristic bands ~1450 cm⁻¹ for Lewis-bound pyridine.
Quantitative Strength Descriptors and Data
The strength of acid sites is quantified through both experimental and computational descriptors. The following table summarizes key quantitative measures relevant for Brønsted/Lewis ratio studies.
Table 1: Quantitative Descriptors of H-ZSM-5 Acid Site Strength
| Descriptor | Brønsted Acid Site (BAS) | Lewis Acid Site (LAS) | Typical Value Range (H-ZSM-5) | Measurement Technique |
|---|---|---|---|---|
| O-H Stretching Frequency (νOH) | Si-OH-Al bridging group | Not Applicable | 3605 - 3610 cm⁻¹ | FT-IR Spectroscopy |
| NH₃ Adsorption Enthalpy (ΔHₐds) | Proton donation to NH₃ | Electron pair acceptance from NH₃ | -120 to -150 kJ/mol (BAS) -100 to -140 kJ/mol (LAS) | Calorimetry, TPD |
| Pyridine IR Band (L-Py) | Band at ~1545 cm⁻¹ (B-Py) | Band at ~1450 cm⁻¹ (L-Py) | -- | FT-IR of Adsorbed Pyridine |
| Deammoniation Temp. (NH₃-TPD) | High-temperature peak | Low to medium-temperature peaks | BAS Peak: 350-450°C | Temperature-Programmed Desorption |
| ¹H NMR Chemical Shift (δH) | Bridging OH proton | Not Applicable | 4.0 - 5.0 ppm (vs. TMS) | Solid-State ¹H MAS NMR |
| 27Al NMR Chemical Shift (δAl) | Tetrahedral framework Al (60-65 ppm) | Octahedral/EFAl (0-10 ppm) | Framework: ~60 ppm EFAl: ~0 ppm | Solid-State ²⁷Al MAS NMR |
| Average Deactivation Energy (Edeact) | Calculated for proton transfer | Calculated for complexation | Varies with reaction | Computational (DFT) |
Table 2: Typical Brønsted/Lewis Ratio Data from Pyridine-IR
| H-ZSM-5 Sample (Si/Al Ratio) | BAS Concentration (μmol/g) | LAS Concentration (μmol/g) | B/L Ratio | Calcination Condition |
|---|---|---|---|---|
| 25 (Parent) | 450 | 50 | 9.0 | 550°C, Air |
| 25 (Steamed) | 320 | 180 | 1.8 | 750°C, 100% H₂O |
| 40 (Parent) | 300 | 30 | 10.0 | 550°C, Air |
| 15 (Parent) | 650 | 70 | 9.3 | 550°C, Air |
Experimental Protocols for Acid Site Characterization
Temperature-Programmed Desorption of Ammonia (NH₃-TPD)
Purpose: To quantify total acid site density and approximate strength distribution. Protocol:
- Pretreatment: ~0.1 g of zeolite is loaded into a quartz U-tube reactor. Activate in situ under helium flow (30 mL/min) by heating to 550°C (5°C/min) and holding for 1 hour.
- Ammonia Saturation: Cool to 100°C. Expose to a stream of 5% NH₃/He (30 mL/min) for 30 minutes.
- Physisorbed NH₃ Removal: Flush with He at 100°C for 1-2 hours to remove weakly bound/physisorbed ammonia.
- Desorption: Heat the sample in He flow (30 mL/min) from 100°C to 700°C at a ramp rate of 10°C/min. Monitor desorbed NH₃ via mass spectrometer (m/z=16) or TCD.
- Analysis: Quantify acid site density by integrating the TPD curve. Peaks at lower (150-300°C) and higher (350-500°C) temperatures are often attributed to weaker (some LAS) and stronger (predominantly BAS) sites, respectively.
Fourier-Transform Infrared Spectroscopy with Pyridine Adsorption (Py-IR)
Purpose: To discriminate and quantify Brønsted and Lewis acid sites. Protocol:
- Wafer Preparation: Press 10-15 mg of zeolite into a self-supporting wafer.
- Pretreatment: Place wafer in a high-temperature IR cell with CaF₂ windows. Evacuate (<10⁻⁴ mbar) and heat to 450°C for 2 hours.
- Background Scan: Cool to 150°C and collect a background spectrum.
- Pyridine Adsorption: Expose the wafer to pyridine vapor (saturated at room temperature) for 5-10 minutes.
- Evacuation: Evacuate at 150°C for 30 minutes to remove physisorbed pyridine.
- Spectrum Acquisition: Collect IR spectrum in the 1400-1700 cm⁻¹ region.
- Quantification: Use molar extinction coefficients (e.g., εB(1545) = 1.13 cm/μmol, εL(1450) = 1.28 cm/μmol) and the integrated area of the bands at ~1545 cm⁻¹ (B-Py) and ~1450 cm⁻¹ (L-Py) to calculate BAS and LAS concentrations.
Visualizing Acidity Pathways and Characterization
Title: Acid Site Characterization Workflow
Title: BAS vs LAS Catalytic Pathways
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Research Reagents for H-ZSM-5 Acid Site Studies
| Item | Function in Research | Technical Specification / Notes |
|---|---|---|
| NH₄-ZSM-5 Zeolite | Parent material. Calcination produces the active H-ZSM-5 (protonic) form. | Defined Si/Al ratio (e.g., 15, 25, 40). Control particle size for diffusion studies. |
| Anhydrous Ammonia (5% in He) | Probe molecule for TPD to quantify total acid site density and strength. | High-purity gas mixture. Requires proper gas handling equipment. |
| Anhydrous Pyridine | Selective IR probe molecule to distinguish and quantify Brønsted vs. Lewis sites. | Must be thoroughly dried (over molecular sieves) before use. Toxic. |
| Deuterated Acetonitrile (CD₃CN) | Weak base IR probe for very strong acid sites; shifts in ν(CN) indicate strength. | Useful for differentiating among strong BAS. |
| Nitrogen (N₂) Gas | Used for adsorption (BET surface area) and as a carrier/purge gas. | Ultra-high purity (99.999%) to prevent contamination. |
| Helium (He) Gas | Inert carrier gas for TPD and pretreatment. | Ultra-high purity. Must be oxygen-free for high-temperature treatments. |
| Calibration Gases (e.g., 1% H₂ in Ar) | For calibrating mass spectrometers or TCD detectors used in TPD/TPR. | Certified standard mixtures. |
| High-Temperature IR Cell | Allows in situ pretreatment and adsorption/desorption studies during IR measurement. | Must have heating capability (>500°C), vacuum, and gas dosing. |
| Quartz Wool & U-Tube Reactors | For packing catalyst samples in flow reactors (TPD, catalysis). | Quartz is inert at high temperatures; pre-clean to remove contaminants. |
| MAS NMR Rotors | For solid-state NMR analysis of ¹H, ²⁷Al, ²⁹Si nuclei to study local structure. | Typically 3.2 or 4 mm zirconia rotors. |
Within the ongoing research paradigm focused on Brønsted-to-Lewis (B/L) acid site ratio comparisons in H-ZSM-5 zeolites, a central thesis has emerged: the precise proportional relationship between these acid site types is not merely a compositional detail but a fundamental descriptor dictating catalytic performance. This whitepaper provides an in-depth technical guide on the intrinsic link between the B/L ratio and core catalytic properties such as activity, selectivity, and deactivation resistance. This relationship is critical for researchers and process chemists, particularly in fields like catalytic bio-oil upgrading and fine chemical synthesis, where H-ZSM-5 is a pivotal material.
Quantitative Data Synthesis: B/L Ratio Impact on Catalytic Metrics
The catalytic outcomes of H-ZSM-5 in model reactions are quantitatively summarized below. Data is synthesized from recent studies on methanol-to-hydrocarbons (MTH), ethanol dehydration, and biomass pyrolysis vapor upgrading.
Table 1: Impact of B/L Ratio on Catalytic Performance in Key Reactions
| Reaction | Typical B/L Ratio Range Studied | Optimum B/L (Approx.) | Key Property Influenced | Observed Trend |
|---|---|---|---|---|
| Methanol to Hydrocarbons (MTH) | 0.5 - 12.0 | 3.0 - 5.0 | Olefin Selectivity (C₂-C₄) | Bell-shaped curve; Moderate B/L maximizes olefins. |
| Ethanol Dehydration to Ethylene | 1.0 - 20.0 | > 8.0 (High B/L) | Ethylene Selectivity (%) | Increases monotonically with higher B/L ratio. |
| Biomass Vapor Deoxygenation | 0.8 - 10.0 | 2.0 - 4.0 | Hydrocarbon Yield & Coke Resistance | Moderate ratios balance deoxygenation and inhibit polycyclic coke. |
| Xylene Isomerization | 2.0 - 15.0 | ~ 10.0 | p-Xylene Selectivity & Catalyst Lifetime | Higher B/L reduces undesirable disproportionation. |
Table 2: Characterization Data for H-ZSM-5 with Modified B/L Ratios
| Modification Method | Total Acidity (mmol NH₃/g) | B/L Ratio (by Py-IR) | Relative Strong Acid Site Density | Mesoporosity (m²/g) |
|---|---|---|---|---|
| Parent H-ZSM-5 (Si/Al=40) | 0.45 | 4.2 | 1.00 (Reference) | 15 |
| Mild Steam Treatment | 0.38 | 1.8 | 0.75 | 18 |
| Mg²⁺ Ion Exchange | 0.41 | 0.7 | 0.65 | 16 |
| Phosphorus Impregnation | 0.35 | 8.5 | 0.60 | 20 |
Experimental Protocols for Key Measurements
Protocol 2.1: Quantifying B/L Ratio via Pyridine Adsorption IR Spectroscopy
- Objective: To discriminate and quantify Brønsted (B) and Lewis (L) acid sites.
- Materials: Vacuum IR cell with KBr windows, pyridine, H-ZSM-5 wafer (∼10 mg/cm²).
- Procedure:
- Pretreat the wafer under vacuum (10⁻² Pa) at 450°C for 2 hours.
- Cool to 150°C and record background spectrum.
- Expose to pyridine vapor (∼1 kPa) for 30 mins.
- Evacuate at 150°C for 1 hour to remove physisorbed pyridine.
- Record IR spectrum in the 1400-1600 cm⁻¹ region.
- Integrate peak areas: Brønsted sites (1545 cm⁻¹, pyridinium ion) and Lewis sites (1455 cm⁻¹, coordinately bonded pyridine).
- Calculate ratio using established molar extinction coefficients (e.g., B: ε₁₅₄₅ = 0.073 cm/μmol, L: ε₁₄₅₅ = 0.100 cm/μmol).
Protocol 2.2: Catalytic Testing in Methanol-to-Hydrocarbons (MTH)
- Objective: To correlate B/L ratio with product selectivity and lifetime.
- Materials: Fixed-bed microreactor, online GC, H-ZSM-5 catalyst (60-80 mesh), methanol feed.
- Procedure:
- Load 100 mg catalyst mixed with SiC diluent.
- Activate in-situ in N₂ flow at 500°C for 1 hour.
- Switch to methanol feed (WHSV = 1.0 h⁻¹) at 370°C.
- Analyze effluent gases and hydrocarbons by online GC every 30 mins.
- Key metrics: Methanol conversion (>99% initial), selectivity to C₂-C₄ olefins, and time-on-stream to 50% conversion (TOS₅₀).
Visualizing Relationships and Workflows
Diagram 1: B/L Ratio Influence on Catalytic Properties
Diagram 2: Py-IR B/L Ratio Measurement Protocol
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Reagents and Materials for B/L Ratio Research
| Item Name | Function / Purpose | Technical Note |
|---|---|---|
| H-ZSM-5 Zeolite (various Si/Al) | Core catalyst material. Si/Al ratio determines total acidity. | Standardize particle size (e.g., 60-80 mesh) for consistent testing. |
| Pyridine (Spectroscopic Grade) | Probing molecule for IR distinction of Brønsted vs. Lewis sites. | Must be thoroughly dried (over molecular sieves) before use. |
| Ammonia (for NH₃-TPD) | Probe for total acid site strength distribution via temperature-programmed desorption. | Use 5% NH₃ in He for safety and dosage control. |
| Inert Gas (Ar, He, N₂) | For catalyst pretreatment, purging, and carrier gas in reactions. | Ultra-high purity (>99.999%) with oxygen/moisture traps. |
| Metal Salt Precursors (e.g., Mg(NO₃)₂) | For ion-exchange to selectively modify Lewis acidity or block sites. | Aqueous solution concentration critical for exchange level. |
| Methanol Feedstock (HPLC Grade) | Reactant for model MTH reaction testing catalytic performance. | Dry to prevent water-induced site masking. |
| Silicon Carbide (SiC) Diluent | Inert material to mix with catalyst bed for improved heat distribution. | Ensure similar mesh size to catalyst to avoid channeling. |
Within the broader thesis on Brønsted to Lewis (B/L) acid site ratio in H-ZSM-5 zeolites, this technical guide examines how post-synthetic conditions dictate catalytic evolution. The B/L ratio is not a static property but a dynamic characteristic shaped by thermal treatment (calcination, steaming) and the reactive chemical environment. Precise control over this ratio is critical for tuning selectivity in hydrocarbon cracking, methanol-to-olefins (MTO) processes, and drug intermediate synthesis, where acid site distribution influences reaction pathways and inhibitor binding.
Mechanisms of Acid Site Evolution
Brønsted acid sites (BAS) are associated with framework aluminum (Al-OH-Si), while Lewis acid sites (LAS) originate from extra-framework aluminum (EFAL) species and framework defects. Processing conditions drive the interconversion between these sites.
- Calcination: The high-temperature treatment in oxygen or air removes organic templates and ammonium ions, generating the protonic H-form. Excessive temperature or duration initiates dealumination, converting BAS (framework Al) to LAS (EFAL).
- Steam Treatment: Hydrothermal conditions accelerate dealumination. Steam hydrolyzes Si-O-Al bonds, extracting aluminum from the framework to form mobile EFAL species, which are strong Lewis acids. This drastically reduces the B/L ratio.
- Reaction Media: During catalysis, the feed (e.g., hydrocarbons, alcohols) can induce in situ changes. Coke deposition preferentially blocks BAS, altering the effective B/L ratio. Certain reactants can also react with or re-integrate EFAL, modifying acid strength and distribution.
Table 1: Impact of Calcination Temperature on H-ZSM-5 (Si/Al=40) Acidic Properties
| Calcination Temperature (°C) | Brønsted Acidity (µmol/g)* | Lewis Acidity (µmol/g)* | B/L Ratio | Primary Effect |
|---|---|---|---|---|
| 550 | 320 | 45 | 7.1 | Standard H-form generation |
| 700 | 280 | 85 | 3.3 | Initial framework dealumination |
| 850 | 150 | 195 | 0.77 | Extensive EFAL formation |
*Data acquired via pyridine FTIR. Values are illustrative from recent literature.
Table 2: Effect of Steam Treatment Severity on Acid Site Population
| Treatment Condition (Temp, Time) | BAS Retention (%) | LAS Increase (vs. parent) | B/L Ratio | Notes |
|---|---|---|---|---|
| Parent (Calcined at 550°C) | 100% | Baseline | 7.1 | Reference |
| 600°C, 2h, 100% H₂O | ~75% | +150% | ~3.2 | Mild dealumination |
| 750°C, 5h, 100% H₂O | ~30% | +400% | ~0.5 | Severe dealumination, mesopore formation |
Table 3: Reaction Media Influence on In Situ B/L Ratio (MTO Process)
| Time-on-Stream (h) | Effective BAS (µmol/g) | Effective LAS (µmol/g) | Apparent B/L Ratio | Cause |
|---|---|---|---|---|
| 0.5 | 320 | 45 | 7.1 | Fresh catalyst |
| 6 | 210 | 90 | 2.3 | Coke selective blocking of BAS |
| 24 | 80 | 110 | 0.73 | Heavy coking & pore blockage |
Experimental Protocols
Protocol 4.1: Controlled Calcination of NH₄-ZSM-5
- Material: 2g of NH₄-ZSM-5 (Si/Al=40) pelletized and sieved to 250-500 µm.
- Setup: Place sample in a quartz tube reactor within a tubular furnace.
- Procedure: Ramp temperature at 2°C/min to target temperature (e.g., 550°C, 700°C, 850°C) under a dry air flow of 50 mL/min. Hold for 5 hours.
- Cooling: Cool to room temperature under dry air flow. Transfer to a desiccator.
- Analysis: Characterize acid sites using in situ pyridine adsorption FTIR or ammonia TPD.
Protocol 4.2: Hydrothermal (Steam) Treatment
- Material: 1g of calcined H-ZSM-5 (from Protocol 4.1, 550°C).
- Setup: Fixed-bed reactor with pre-heater for water vaporization. Place catalyst in isothermal zone.
- Procedure: Pass N₂ (50 mL/min) through a saturator containing deionized water at 70°C (to achieve partial pressure). Heat reactor to treatment temperature (e.g., 600°C or 750°C). Maintain conditions for 2-5 hours.
- Quench: Switch to dry N₂ flow, cool rapidly to room temperature.
- Analysis: Use ²⁷Al MAS NMR to quantify framework vs. extra-framework Al, and FTIR for acid typing.
Protocol 4.3: Probing Acid Sites via In Situ Pyridine FTIR
- Material: 20 mg of treated zeolite pressed into a self-supported wafer.
- Setup: Load wafer into a high-temperature IR cell with CaF₂ windows, capable of vacuum and gas flow.
- Dehydration: Evacuate cell (<10⁻³ mbar) and heat to 400°C for 1 hour. Cool to 150°C for adsorption.
- Adsorption: Expose wafer to pyridine vapor (equilibrium pressure ~5 mbar) for 15 min.
- Desorption: Evacuate at 150°C for 30 min to remove physisorbed pyridine.
- Measurement: Record IR spectrum. Quantify BAS (band ~1545 cm⁻¹) and LAS (band ~1455 cm⁻¹) using established extinction coefficients (e.g., E(BAS) ≈ 1.67 cm/µmol, E(LAS) ≈ 2.22 cm/µmol).
Visualizations
Title: Pathways of Zeolite Acid Site Evolution
Title: Experimental Workflow for B/L Ratio Study
Title: Interconversion of Acid Sites
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 4: Key Research Reagent Solutions for B/L Ratio Studies
| Item | Function/Explanation |
|---|---|
| NH₄-ZSM-5 Zeolite (various Si/Al) | The precursor material. Ion exchange with ammonium provides the source for protonic Brønsted sites upon calcination. Si/Al ratio determines total acid capacity. |
| Pyridine (Spectroscopic Grade) | A probe molecule for FTIR spectroscopy. Selectively chemisorbs to BAS (pyridinium ion) and LAS (coordinately bound pyridine), allowing quantification. |
| Deionized Water (Ultra-high Purity) | Used for generating steam in hydrothermal treatments. Purity is critical to avoid contamination by metal ions that could alter dealumination kinetics. |
| High-Purity Gases (O₂, N₂, Air) | Essential for calcination (O₂, air) and as inert carriers (N₂) during treatment and cooling. Moisture traps are required to maintain dry conditions. |
| Reference Catalysts (e.g., ALFA Zeolites) | Well-characterized commercial zeolites (e.g., H-ZSM-5, H-Y) used as benchmarks for validating analytical methods and experimental setups. |
| Inert Sieve Material (Quartz Wool/Beads) | Used to support catalyst beds in fixed-bed reactors, ensuring proper gas flow and temperature distribution during treatments and catalysis. |
| FTIR Extinction Coefficients (for Pyridine) | Calibrated constants (e.g., ε₁₅₄₅, ε₁₄₅₅) necessary to convert IR band intensities to quantitative acid site densities (µmol/g). |
Measuring & Modulating: Advanced Techniques to Characterize and Engineer the B/L Acid Ratio
Within the context of Brønsted to Lewis (B/L) acid site ratio research on H-ZSM-5 zeolites, accurate spectroscopic quantification is paramount. This guide details best practices for Fourier-Transform Infrared (FTIR) spectroscopy, Pyridine-probed IR (Py-IR), and Nuclear Magnetic Resonance (NMR) spectroscopy, which are cornerstone techniques for differentiating and quantifying acid site types. These methods provide critical insights into catalyst performance, directly impacting fields from petrochemical refining to drug intermediate synthesis.
Core Principles of Acid Site Quantification
Brønsted (B) acid sites in H-ZSM-5 are associated with bridging hydroxyl groups (e.g., Si-OH-Al), while Lewis (L) acid sites are associated with electron-deficient aluminum species. The B/L ratio significantly influences catalytic activity, selectivity, and deactivation behavior in reactions such as cracking, isomerization, and alkylation.
Fourier-Transform Infrared (FTIR) Spectroscopy
Experimental Protocol for Hydroxyl Region Analysis
- Sample Preparation: Press zeolite powder (≈20 mg) into a self-supporting wafer (10-20 mg/cm²). Activate in a dedicated IR cell with heating capability under high vacuum (≤10⁻⁵ mbar) at 450-500°C for 1-2 hours to remove adsorbed water and contaminants.
- Data Acquisition: Cool to analysis temperature (typically 150°C or room temperature). Collect spectra in transmission mode using a high-sensitivity MCT detector. Parameters: resolution 4 cm⁻¹, 64-128 scans.
- Quantification: The band at ~3605 cm⁻¹ is assigned to Brønsted acid site hydroxyls (bridging OH). The band at ~3745 cm⁻¹ is assigned to non-acidic terminal silanols. Baseline correction and peak deconvolution (using Gaussian/Lorentzian profiles) are essential. The integrated absorbance area, combined with the sample wafer mass and area, can be used with the Beer-Lambert law and known extinction coefficients for semi-quantitative comparison.
Pyridine-Adsorbed FTIR (Py-IR) for B/L Discrimination
Detailed Experimental Protocol
- Activation: Follow the FTIR wafer activation protocol above.
- Pyridine Adsorption: Expose the activated wafer to pyridine vapor (saturated at room temperature) for 5-10 minutes. Physisorbed pyridine is then removed by evacuating at 150°C for 30-60 minutes.
- Spectra Acquisition: Collect spectrum after evacuation.
- Quantification Analysis: Analyze the region 1400-1600 cm⁻¹.
- Brønsted-bound pyridine (PyH⁺): Characteristic bands at ~1545 cm⁻¹ and ~1630 cm⁻¹.
- Lewis-bound pyridine (PyL): Characteristic bands at ~1455 cm⁻¹ and ~1620 cm⁻¹.
- The integrated areas of the 1545 cm⁻¹ (B) and 1455 cm⁻¹ (L) bands are used for quantification.
- Calculation: Acid site concentrations (μmol/g) are calculated using the formula:
C = (A * S) / (ε * m)where C = concentration, A = integrated band area (cm⁻¹), S = wafer area (cm²), m = wafer mass (g), and ε = molar extinction coefficient (cm/μmol). Critical Note: Published ε values vary. Consistent use of internally calibrated or carefully selected literature values (e.g., εB ~1.67 cm/μmol, εL ~2.22 cm/μmol for Py-IR on zeolites) is mandatory for comparability. The B/L ratio is then the simple quotient CB / CL.
Table 1: Key FTIR/Py-IR Band Assignments for H-ZSM-5
| Vibration Mode | Wavenumber (cm⁻¹) | Assignment |
|---|---|---|
| O-H Stretching | ~3605 | Brønsted Acid Site (bridging Si-OH-Al) |
| O-H Stretching | ~3745 | Terminal Silanol (Si-OH), non-acidic |
| Pyridine Ring Vibration | ~1545 | Pyridinium Ion (PyH⁺, Brønsted site) |
| Pyridine Ring Vibration | ~1455 | Coordinated Pyridine (PyL, Lewis site) |
| Pyridine Ring Vibration | ~1490 | Overlap Band (B + L contribution) |
Table 2: Typical Quantitative B/L Data from Py-IR on H-ZSM-5
| H-ZSM-5 Sample (Si/Al) | Brønsted Acidity (μmol/g) | Lewis Acidity (μmol/g) | B/L Ratio | Evac. Temp. |
|---|---|---|---|---|
| 25 | 350 - 450 | 80 - 120 | 3.5 - 5.5 | 150°C |
| 40 | 250 - 320 | 50 - 80 | 4.5 - 6.5 | 150°C |
| 200 | 50 - 80 | 15 - 25 | 3.0 - 4.5 | 150°C |
Nuclear Magnetic Resonance (NMR) Spectroscopy
Experimental Protocol for ¹H MAS NMR
- Sample Preparation: Load activated zeolite powder into a magic-angle spinning (MAS) rotor in a dry, inert atmosphere (glovebox) to prevent rehydration.
- Data Acquisition: Acquire ¹H MAS NMR spectra at high spinning speeds (≥10 kHz) to minimize interference. Use a single-pulse or spin-echo sequence. Reference to TMS (0 ppm) via a secondary solid reference like adamantane.
- Quantification: The signal at ~4.3 ppm is assigned to bridging OH (Brønsted sites). The signal at ~1.8-2.0 ppm is assigned to extra-framework Al-OH (Lewis-associated). Signal at ~0.9-1.2 ppm may be assigned to non-acidic Al-OH species. Deconvolution and integration of these peaks provide a direct quantitative B/L ratio without reliance on probe molecules or extinction coefficients.
Experimental Protocol for ²⁷Al MAS NMR
- Sample Preparation: Similar to ¹H NMR, ensure dry handling.
- Data Acquisition: Acquire ²⁷Al MAS NMR at high spinning speeds. Use very short pulses to quantitatively excite all Al nuclei, including the distorted, often invisible, Lewis sites.
- Quantification: The peak at ~55 ppm is assigned to tetrahedral framework Al (origin of Brønsted sites). Peaks at ~0 ppm and ~30 ppm are assigned to octahedral and pentacoordinated extra-framework Al (EFAl), respectively, which are associated with Lewis acidity. Quantification requires careful integration and consideration of the often "invisible" Al, typically mitigated by using a very low pulse angle or isotopic enrichment.
Table 3: NMR Chemical Shift References for H-ZSM-5 Acid Sites
| Nucleus | δ (ppm) | Assignment | Relation to Acidity |
|---|---|---|---|
| ¹H | 4.2 - 4.5 | Bridging Si-OH-Al | Brønsted Site |
| ¹H | 1.8 - 2.0 | OH on Extra-framework Al | Lewis Site Associated |
| ¹H | ~0.9 - 1.2 | Non-acidic Al-OH | Lewis Site Associated |
| ²⁷Al | ~55 | Tetrahedral Al (Framework) | Brønsted Site Precursor |
| ²⁷Al | ~0, ~30 | Octahedral/Penta Al (EFAl) | Lewis Site |
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for Spectroscopic B/L Analysis
| Item | Function & Critical Notes |
|---|---|
| H-ZSM-5 Zeolites | Study material with varying Si/Al ratios (e.g., 15, 25, 40, 200). Must be pre-activated. |
| Pyridine, ≥99.9% | IR probe molecule. Must be thoroughly dried over molecular sieves and distilled under inert atmosphere to prevent water contamination. |
| Deuterated NMR Solvents (e.g., CDCl₃, D₂O) | For locking and referencing in solution-state NMR of extracted samples or probe molecule complexes. |
| High-Vacuum IR Cell | Equipped with KBr or CaF₂ windows, heating jacket, and gas/vapor dosing system. Essential for in situ activation and probing. |
| MAS NMR Rotors | Zirconia rotors (3.2 mm or 4 mm) with gas-tight caps for solid-state NMR. |
| Reference Materials | Adamantane (for ¹H NMR referencing), Al(NO₃)₃ solution (for ²⁷Al NMR referencing), and certified KBr pellets (for FTIR background). |
| Dry-Air/N₂ Glovebox | For moisture-free handling of activated zeolites during NMR rotor packing and wafer transfer. |
Experimental Workflow & Data Synthesis
Title: Workflow for Spectroscopic B/L Ratio Analysis on H-ZSM-5
- Complementary Techniques: Py-IR and NMR should be used in tandem. Py-IR probes accessible sites for a specific molecule, while ¹H NMR provides a more direct, probe-independent count of hydroxyl types.
- Extinction Coefficients: The largest source of error in Py-IR is the ε value. Use internally calibrated values for highest accuracy.
- Sample Integrity: Strict control of activation conditions and prevention of atmospheric rehydration are non-negotiable for reproducible results.
- Data Reporting: Always report full experimental details: activation temperature/time, pyridine evacuation temperature, NMR spinning speeds, pulse conditions, and all quantification parameters. This enables meaningful comparison within the research community focused on H-ZSM-5 B/L ratios.
This technical guide details the application of Temperature-Programmed Desorption (TPD) and Calorimetry to characterize acid strength distribution in zeolites. The methodologies described herein are framed within a comprehensive research thesis aimed at quantifying and comparing the Brønsted (B) to Lewis (L) acid site ratio in H-ZSM-5 catalysts. Precise determination of acid strength distribution is critical for understanding catalytic performance in hydrocarbon conversion, a key interest for researchers in catalysis and pharmaceutical intermediate synthesis.
Core Principles and Techniques
Temperature-Programmed Desorption (TPD) of Probe Molecules
TPD measures the temperature-dependent desorption of basic probe molecules (e.g., NH₃, pyridine) pre-adsorbed on acid sites. Stronger acid sites retain probe molecules more tightly, leading to desorption at higher temperatures. The resulting spectrum (desorption rate vs. temperature) is deconvoluted to quantify site concentration and strength.
Calorimetry of Probe Molecule Adsorption
Microcalorimetry directly measures the heat released upon the differential adsorption of a probe molecule. The integral heat vs. coverage profile provides a direct, quantitative measure of acid site strength distribution, with stronger sites yielding higher heats of adsorption.
Experimental Protocols
NH₃-TPD Protocol for H-ZSM-5
- Pre-treatment: Load ~100 mg of H-ZSM-5 pellet (180-250 µm) into a quartz U-tube reactor. Heat to 500°C (5°C/min) under He flow (30 mL/min) for 2 hours to clean the surface.
- Saturation: Cool to 100°C. Switch to a 5% NH₃/He stream (30 mL/min) for 60 minutes to ensure saturation of acid sites.
- Physisorbed NH₃ Removal: Switch back to pure He (30 mL/min) at 100°C for 90-120 minutes to remove weakly physisorbed ammonia.
- Desorption: Heat the sample to 700°C at a constant rate (typically 10°C/min) under He flow. Monitor desorbed NH₃ using a thermal conductivity detector (TCD) or mass spectrometer (MS).
- Quantification: Calibrate the TCD signal using known pulses of NH₃. The total acid site density is calculated from the integrated desorption peak area.
Differential Adsorption Microcalorimetry Protocol
- Sample Activation: Activate a fresh sample (~100 mg) in the calorimetry cell under vacuum (<10⁻⁵ mbar) at 450°C for 4 hours.
- Calorimeter Equilibration: Cool the sample to the adsorption temperature (commonly 150°C). Allow the calorimeter and sensitive thermopiles to reach thermal equilibrium.
- Dosed Adsorption: Introduce small, successive doses of probe vapor (e.g., ammonia, pyridine) into the cell. After each dose, measure the evolved heat until equilibrium pressure is stable.
- Data Acquisition: Record the equilibrium pressure and the corresponding integral heat for each dose. Continue until no further heat is detected upon dosing (surface saturation).
- Analysis: Plot the differential heat of adsorption (kJ/mol) vs. coverage (µmol/g). The curve's shape reveals the strength distribution.
Table 1: Characteristic Acid Strength Data for H-ZSM-5 from Literature
| Probe Molecule | Technique | Peak Desorption Temp. / Heat Range | Assigned Site Type | Typical Site Density (µmol/g) |
|---|---|---|---|---|
| Ammonia (NH₃) | NH₃-TPD | Low Temp.: ~200°C | Weak Acid Sites | 100-300 |
| High Temp.: ~400°C | Strong Brønsted Sites | 200-600 | ||
| Ammonia (NH₃) | Calorimetry | Initial Heat: 140-160 kJ/mol | Strong Brønsted Sites | Varies with Si/Al |
| Final Heat: <80 kJ/mol | Weak/Lewis Sites | |||
| Pyridine (Py) | IR + TPD | ~150°C | Lewis-bound Py | For B/L Ratio |
| ~450°C | Brønsted-bound Py | |||
| Note: Values are representative and depend on H-ZSM-5 Si/Al ratio, preparation, and pre-treatment history. |
Table 2: B/L Ratio Determination via Complementary Techniques
| Method | Probe Used | Measurement Principle | B/L Ratio Output | Key Advantage |
|---|---|---|---|---|
| Pyridine FTIR | Pyridine | IR bands at ~1545 cm⁻¹ (B) and ~1450 cm⁻¹ (L) | Quantitative via extinction coeff. | Direct speciation |
| NH₃-TPD + Py-IR | NH₃ & Py | TPD gives total sites; IR gives B/L fraction | Calculated | Separates strength & type |
| Adsorption Calorimetry | NH₃ or Py | Heat distribution profiles deconvolution | Inferred from strength maps | Direct strength measure |
Visualizing Workflows and Relationships
Title: TPD Experimental Sequence for Acid Site Analysis
Title: Integrated Approach for B/L Ratio Thesis Research
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Acid Site Characterization Experiments
| Item | Function in Experiment | Technical Notes |
|---|---|---|
| H-ZSM-5 Zeolite (various Si/Al) | Primary catalyst sample under investigation. | Si/Al ratio (e.g., 15, 25, 40) determines total acid site density. |
| Anhydrous Ammonia (5% in He) | Standard basic probe molecule for TPD/Calorimetry. | Interacts with both Brønsted and Lewis sites. |
| Anhydrous Pyridine | Sterically demanding probe; used in IR for B/L distinction. | Can be dosed via vapor phase or from a saturated He stream. |
| Ultra-High Purity Helium | Carrier gas for TPD; purge and activation gas. | Must be further dried and oxygen-trapped. |
| Quartz Wool & U-tube Reactor | Holds catalyst bed in flow system. | Inert at high temperatures. |
| Micromeritics ASAP 2020 or Calorimeter | Commercial instrument for precise gas adsorption & calorimetry. | Equipped with a precision dosing system and calorimetric cell. |
| FTIR Spectrometer with DRIFTS/HVC | For in situ identification of acid site type (B vs. L). | Must have a controlled environment cell for high-temp/vacuum. |
| Thermal Conductivity Detector (TCD) | Standard detector for quantifying desorbed NH₃ in TPD. | Requires careful calibration with known NH₃ volumes. |
This whitepaper details targeted synthetic strategies to modulate the Brønsted (B) to Lewis (L) acid site ratio in H-ZSM-5 zeolites. This control is central to a broader thesis investigating the catalytic and adsorptive performance of ZSM-5 in hydrocarbon conversion and drug precursor synthesis. Precise B/L ratio manipulation directs selectivity in key reactions such as methanol-to-hydrocarbons (MTH) and biomass upgrading, which are critical for developing efficient, sustainable pharmaceutical manufacturing pathways.
Foundational Principles of Acidity in ZSM-5
Brønsted acid sites originate from bridging hydroxyl groups (Si-OH-Al), while Lewis acid sites arise from extra-framework aluminum (EFAl), coordinatively unsaturated Al species, or framework defects. The B/L ratio profoundly influences reaction mechanisms: a high B/L favors reactions requiring strong proton donation (e.g., cracking, isomerization), while a balanced or high L content can facilitate dehydrogenation and condensation.
Core Synthetic Levers for B/L Ratio Control
Framework Si/Al Ratio
The bulk Si/Al ratio is the primary determinant of total Brønsted acidity. Higher Si/Al decreases the density of framework Al, thus reducing the maximum possible Brønsted sites. However, synthesis conditions and subsequent treatments dictate how much framework Al converts to EFAl (Lewis sites).
Table 1: Impact of Gel Si/Al on Final B/L Ratio in H-ZSM-5
| Gel Si/Al Ratio | Typical Synthesis Template | Resultant H-ZSM-5 B Acid Density (μmol/g) | Typical B/L Ratio (from Py-IR) | Dominant Acid Type |
|---|---|---|---|---|
| 15 | Tetrapropylammonium (TPAOH) | ~450 | 3.5 - 4.5 | Brønsted-dominant |
| 40 | Tetrapropylammonium (TPAOH) | ~180 | 1.8 - 2.5 | Balanced |
| 100 | Tetrapropylammonium (TPAOH) | ~80 | 0.8 - 1.5 | Lewis-enhanced |
| 200 | Mixed Template (TPABr/NaOH) | ~40 | 0.5 - 1.0 | Lewis-dominant |
Organic Structure-Directing Agents (Templates)
The choice of template influences crystal size, morphology, and Al distribution (framework vs. extra-framework), thereby affecting the B/L ratio.
Experimental Protocol: Synthesis of ZSM-5 with Different Templates
- Gel Preparation: For a target Si/Al=40, mix sodium aluminate (NaAlO₂) and fumed silica (SiO₂) in deionized water.
- Template Addition: Divide the gel into four parts. Add an equimolar amount of:
- Part A: Tetrapropylammonium hydroxide (TPAOH, 1.0 M aqueous).
- Part B: Tetrabutylammonium hydroxide (TBAOH).
- Part C: Hexamethylenediamine (HDA).
- Part D: A mixture of TPAOH and dimethyloctylamine (DMOA).
- Hydrothermal Synthesis: Stir each gel for 24h, then transfer to Teflon-lined autoclaves. Crystallize at 170°C for 48-72h.
- Post-treatment: Filter, wash, dry at 110°C, and calcine at 550°C for 6h to remove the template.
- Ion Exchange: Convert Na-ZSM-5 to H-ZSM-5 via three exchanges with 1M NH₄NO₃ solution (80°C, 2h), followed by drying and calcining at 500°C for 4h.
- Characterization: Analyze B/L ratio via Pyridine-adsorbed FTIR (Py-IR) at 150°C (Brønsted: 1545 cm⁻¹; Lewis: 1455 cm⁻¹).
Table 2: Effect of Organic Template on B/L Ratio (Si/Al Gel = 40)
| Template System | Crystal Size (μm) | B Acid Site Density (μmol/g) | B/L Ratio (Py-IR) | Notes |
|---|---|---|---|---|
| TPAOH | 0.5 - 1.0 | 175 | 2.1 | Standard, uniform crystals |
| TBAOH | 2.0 - 5.0 | 165 | 1.5 | Larger crystals, more internal defects (L) |
| HDA | 0.1 - 0.3 (Nano) | 160 | 0.9 | High external surface, significant EFAl |
| TPAOH/DMOA | 0.2 - 0.5 | 170 | 1.8 | Hierarchical pores, moderate L increase |
Post-Synthetic Ion Exchange and Modification
Controlled ion exchange and dealumination are critical for fine-tuning the B/L ratio post-synthesis.
Experimental Protocol: Controlled Ion Exchange/Dealumination for B/L Adjustment Protocol A: Steam Dealumination (Increases Lewis Acidity)
- Place H-ZSM-5 (Si/Al=40) in a quartz tube reactor.
- Pass 100% steam at 500-700°C for 1-5 hours.
- The steam selectively removes framework Al, creating EFAl (Lewis sites) and mesopores.
Protocol B: Mild Acid Washing (Selective EFAl Removal)
- After steam treatment, treat the zeolite with 0.1M HNO₃ or 0.1M (NH₄)₂H₂EDTA at 80°C for 2h.
- This dissolves EFAl, reducing Lewis sites and increasing the relative B/L ratio.
Protocol C: Ion Exchange with Hydrolyzing Cations (e.g., La³⁺)
- Stir H-ZSM-5 in 0.1M La(NO₃)₃ solution at 80°C for 6h.
- Filter, wash, dry, and calcine at 550°C.
- La³⁺ hydrolyzes to form [La(OH)]²⁺, which occupies cation sites and generates new, strong Lewis acid sites.
Table 3: Impact of Post-Synthetic Treatments on B/L Ratio
| Starting Material | Treatment Condition | Resultant B Acid (μmol/g) | Resultant L Acid (μmol/g) | B/L Ratio | Process Effect |
|---|---|---|---|---|---|
| H-ZSM-5 (Si/Al=40) | None (Ref.) | 180 | 85 | 2.1 | Baseline |
| H-ZSM-5 (Si/Al=40) | Steam, 600°C, 2h | 120 | 155 | 0.77 | Creates EFAl (L) |
| Steam-treated ZSM-5 | 0.1M HNO₃, 80°C, 2h | 115 | 95 | 1.2 | Removes some EFAl |
| H-ZSM-5 (Si/Al=40) | 0.1M La³⁺ Exchange | 175 | 210 | 0.83 | Adds cationic L sites |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 4: Key Reagents for ZSM-5 Synthesis and B/L Ratio Analysis
| Reagent/Material | Function & Role in B/L Control | Notes |
|---|---|---|
| Fumed Silica (SiO₂) | High-purity silica source. Determines final Si content. | Reactivity influences crystallization rate and Al incorporation. |
| Sodium Aluminate (NaAlO₂) | Aluminum source. Precise control defines maximum framework Al sites. | Must be fresh to avoid hydrolysis and inconsistent Al feed. |
| Tetrapropylammonium Hydroxide (TPAOH) | Structure-directing agent (SDA) and alkali source. Directs MFI structure. | Concentration and purity affect crystal size and Al distribution. |
| Ammonium Nitrate (NH₄NO₃) | For ion exchange to convert Na-ZSM-5 to H-ZSM-5. | Multiple exchanges are necessary for complete protonation. |
| Pyridine (anhydrous) | Probe molecule for FTIR spectroscopy to quantify B and L sites. | Must be thoroughly dried. Spectra analyzed at different temperatures. |
| Lanthanum(III) Nitrate (La(NO₃)₃·6H₂O) | Source of hydrolyzing cation for introducing tailored Lewis acidity. | Calcination after exchange is crucial to form active [La(OH)]²⁺ species. |
| Ethylenediaminetetraacetic Acid (EDTA) Salts | Chelating agent for selective removal of extra-framework Al (EFAl). | Mild dealumination agent; can increase relative B/L ratio. |
Visualization of Synthesis Strategy and Acid Site Analysis Workflow
Title: Workflow for Synthesizing H-ZSM-5 with Controlled B/L Ratio
Title: Generation Pathways for Brønsted and Lewis Acid Sites
Strategic control of the B/L acid site ratio in H-ZSM-5 is a multi-parameter synthesis and post-synthesis optimization problem. The gel Si/Al ratio sets the theoretical ceiling for Brønsted sites, the organic template directs Al siting and crystal morphology, and ion exchange/steam treatments provide precise final tuning. Integrating these levers, as detailed in the protocols and data tables, enables researchers to tailor H-ZSM-5 catalysts for specific mechanistic pathways in hydrocarbon and pharmaceutical precursor chemistry, directly testing hypotheses within the broader Brønsted to Lewis acid site comparison research thesis.
This whitepaper provides an in-depth technical guide to three critical post-synthetic modification techniques for tailoring the Brønsted (BAS) to Lewis (LAS) acid site ratio in H-ZSM-5 zeolites. Operating within the context of advanced research on acid site ratio comparisons, we detail methodologies, quantitative outcomes, and practical protocols for dealumination, isomorphous substitution, and metal impregnation. These techniques are pivotal for optimizing catalyst performance in petrochemical and pharmaceutical precursor synthesis.
The catalytic performance of H-ZSM-5 in reactions such as cracking, isomerization, and the methanol-to-hydrocarbons process is governed by the nature, strength, and density of its acid sites. Brønsted acid sites (BAS), originating from bridging hydroxyl groups (Si-OH-Al), and Lewis acid sites (LAS), arising from extra-framework aluminum (EFAL) or introduced metal cations, play synergistic yet distinct roles. The precise BAS/LAS ratio is a critical determinant of activity, selectivity, and catalyst lifetime. Post-synthetic modification allows for precise engineering of this ratio beyond the constraints of direct synthesis.
Chapter 1: Dealumination - Selective Removal for Ratio Tuning
Dealumination involves the partial removal of framework aluminum from the zeolite, directly reducing BAS count and often generating EFAL, which acts as LAS.
Core Protocols
1.1.1 Steam Dealumination
- Method: Place H-ZSM-5 (SiO₂/Al₂O₃ ratio 30-80) in a quartz reactor. Treat with 100% steam (0.5-1.0 bar partial pressure) at 500-700°C for 1-6 hours under atmospheric pressure. Follow with a mild acid wash (e.g., 0.1M HNO₃, 80°C, 2h) to remove dislodged EFAL if a purer LAS source is desired.
- Key Variables: Temperature, steam partial pressure, duration.
1.1.2 Acid Dealumination
- Method: Reflux H-ZSM-5 (typically 1g/50ml) in a mineral acid solution (e.g., 0.1-6M HCl or HNO₃) at 80-100°C for 1-24 hours. Filter, wash thoroughly with deionized water, and dry at 110°C overnight.
- Key Variables: Acid type, concentration, temperature, time.
Table 1: Impact of Dealumination Methods on H-ZSM-5 Acid Site Properties
| Modification Method | Condition Example | Parent SiO₂/Al₂O₃ | Resultant SiO₂/Al₂O₃ | BAS Density (µmol/g)* | LAS Density (µmol/g)* | BAS/LAS Ratio | Key Effect |
|---|---|---|---|---|---|---|---|
| Steam | 600°C, 4h, 1 bar H₂O | 40 | 110 | 180 | 95 | 1.9 | High LAS from EFAL, mesoporosity creation. |
| Steam + Acid Wash | 600°C, 4h + 0.1M HNO₃ | 40 | 105 | 160 | 45 | 3.6 | Removes some EFAL, higher BAS/LAS. |
| Mild Acid | 0.5M HCl, reflux, 4h | 40 | 75 | 250 | 60 | 4.2 | Moderate BAS reduction, low EFAL. |
| Severe Acid | 6M HCl, reflux, 24h | 40 | 200 | 85 | 25 | 3.4 | High BAS removal, low total acidity. |
Representative values from literature; measured via NH₃-TPD and pyridine FTIR.
Dealumination Workflow
Title: Dealumination Pathways for Modifying Acid Sites in H-ZSM-5
Chapter 2: Isomorphous Substitution - Framework Integration
This method replaces framework aluminum with other tri- or tetravalent elements (e.g., Fe, Ga, B), altering acid strength and type.
Core Protocol: Framework Iron Substitution (Aqueous Route)
- Preparation of Solution: Dissolve an appropriate amount of Fe(NO₃)₃·9H₂O in deionized water to achieve a target Fe/Al atomic ratio (e.g., 0.1-0.5). Use a liquid-to-solid ratio of 50 ml/g zeolite.
- Ion Exchange: Add H-ZSM-5 to the solution and stir at 80°C for 6 hours. Filter and wash.
- Calcination & Hydrothermal Treatment: Dry the solid at 110°C, then calcine in air at 550°C for 5 hours. Optionally, a mild steam treatment (e.g., 500°C, 2h) can facilitate Fe migration into the framework.
- Key Variables: Precursor type, concentration, temperature, and post-exchange treatment.
Table 2: Impact of Isomorphous Substitution on H-ZSM-5 Acid Properties
| Incorporated Element | Precursor | Treatment | BAS Strength* | LAS Density* | BAS/LAS Ratio | Notable Change |
|---|---|---|---|---|---|---|
| Boron (B³⁺) | H₃BO₃ | Calcination @ 550°C | Weakened | Slight Increase | ~5-8 | Creates very weak BAS, lowers activity. |
| Iron (Fe³⁺) | Fe(NO₃)₃ | Steaming @ 500°C | Moderately Weakened | Significantly Increased | ~1-3 | Introduces strong redox LAS, bifunctional catalysis. |
| Gallium (Ga³⁺) | Ga(NO₃)₃ | Calcination @ 600°C | Slightly Weakened | Increased | ~2-4 | Promotes dehydrogenation activity. |
Relative comparison to parent H-ZSM-5; measured by pyridine/2,6-di-tert-butylpyridine FTIR and NH₃-TPD.
Isomorphous Substitution Logic
Title: Mechanism of Isomorphous Substitution in H-ZSM-5
Chapter 3: Metal Impregnation - Surface Deposition for Bifunctionality
Impregnation deposits metal cations (e.g., Zn, Ag, Cu, La) onto the zeolite, primarily introducing new LAS and/or blocking BAS.
Core Protocol: Incipient Wetness Impregnation of Zinc
- Pore Volume Determination: Prior to impregnation, determine the water-absorbing pore volume of the H-ZSM-5 (typically ~0.4-0.8 ml/g) by gradual addition of water to a dry sample until saturation.
- Solution Preparation: Dissolve Zn(NO₃)₂·6H₂O in deionized water equal to the determined pore volume to achieve a target Zn loading (e.g., 1-5 wt.%).
- Impregnation: Add the solution dropwise to the zeolite powder with vigorous mixing to ensure uniform distribution.
- Drying & Calcination: Age the wet solid at room temperature for 2-4 hours, dry at 110°C overnight, and calcine in air at 500°C for 4 hours to decompose the nitrate.
Table 3: Impact of Metal Impregnation on H-ZSM-5 Acid Site Ratio
| Impregnated Metal | Loading (wt.%) | Primary Effect on BAS | New LAS Type | Typical BAS/LAS Post-Impregnation | Primary Function |
|---|---|---|---|---|---|
| Zinc (Zn²⁺) | 2.0 | Partial Neutralization | Strong Lewis (Zn²⁺) | 0.5 - 1.5 | Ethane aromatization, dehydrogenation. |
| Lanthanum (La³⁺) | 3.0 | Stabilization/Blocking | Moderate Lewis (La³⁺) | 1.0 - 2.0 | BAS stabilization, reduces deactivation. |
| Silver (Ag⁺) | 1.5 | Partial Exchange | Weak Lewis (Ag⁺) | 2.0 - 3.0 | Selective catalytic reduction, olefin separation. |
| Copper (Cu²⁺) | 4.0 | Significant Neutralization | Redox Lewis (Cu²⁺/Cu⁺) | 0.3 - 1.0 | NOx reduction, methane oxidation. |
Metal Impregnation Workflow
Title: Incipient Wetness Impregnation Process for H-ZSM-5
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 4: Key Reagent Solutions and Materials for Post-Synthetic Modifications
| Item | Typical Specification/Example | Primary Function in Research |
|---|---|---|
| H-ZSM-5 Zeolite | SiO₂/Al₂O₃ = 30, 50, 80; NH₄⁺ or H⁺ form. | The foundational catalyst material for all modifications. |
| Steam Generator | Precision oven with steam inlet, or dedicated steam calciner. | Provides controlled hydrothermal environment for dealumination. |
| Mineral Acids | HCl (37%), HNO₃ (69%), Analytical Grade. | Agent for acid dealumination or washing to remove EFAL. |
| Metal Salt Precursors | Fe(NO₃)₃·9H₂O, Ga(NO₃)₃, Zn(NO₃)₂·6H₂O, (NH₄)₂SO₄, 99% purity. | Source of metal cations for substitution or impregnation. |
| Tube Furnace / Calciner | Programmable up to 900°C, with controlled atmosphere (air, N₂). | For calcination, thermal activation, and steam treatments. |
| Ammonia (NH₃) Gas | 5% in He or pure NH₃, for Temperature-Programmed Desorption (TPD). | Probe molecule for quantifying total acid site density. |
| Pyridine | Anhydrous, 99.8%, spectroscopic grade. | FTIR probe to distinguish and quantify BAS vs. LAS. |
| Nitrogen Physisorption | N₂ at 77K, using BET/BJH analysis instruments. | Characterizes surface area, micropore, and mesopore volume changes. |
The strategic application of dealumination, isomorphous substitution, and metal impregnation provides a powerful triad for mastering the BAS/LAS ratio in H-ZSM-5. Dealumination offers a direct route to reduce BAS and generate EFAL-LAS. Isomorphous substitution modifies the framework itself, tuning acid strength and incorporating redox-active LAS. Metal impregnation adds distinct, often strongly Lewis-acidic, functionalities. The choice of technique depends on the target reaction: a low BAS/LAS ratio (via Zn impregnation) favors dehydrogenation, while a stabilized, moderate ratio (via La or mild steam dealumination) may enhance cracking selectivity and longevity. Continued research quantifying the precise ratio-activity-selectivity relationships is essential for the rational design of next-generation zeolite catalysts in both petrochemical and fine chemical synthesis.
This whitepaper presents an in-depth technical guide on optimizing the Brønsted-to-Lewis (B/L) acid site ratio in H-ZSM-5 zeolites for three critical catalytic processes: Methanol-to-Olefins (MTO), Hydrocracking, and Xylene Isomerization. The content is framed within a broader thesis investigating the strategic manipulation of acid site distributions in zeolitic frameworks to enhance selectivity, activity, and catalyst longevity. The interplay between Brønsted (proton-donating) and Lewis (electron-accepting) sites dictates reaction pathways, coke formation, and product distribution, making the B/L ratio a pivotal parameter for industrial catalyst design.
Table 1: Optimal B/L Ratios and Performance Outcomes for Target Reactions
| Reaction System | Optimal B/L Ratio Range (Measured by Py-IR) | Key Performance Metric at Optimum | Typical Catalyst Modifications | Reference Year |
|---|---|---|---|---|
| Methanol-to-Olefins (MTO) | 3.5 – 5.2 | Ethylene+Propylene Selectivity > 80%, Lifetime > 300 h | Mg, P, Si deposition; Steaming | 2023 |
| Heavy Feed Hydrocracking | 1.8 – 2.5 | Middle Distillate Yield > 65%, Reduced Gas Coke | NiMo, NiW impregnation; controlled dealumination | 2022 |
| Xylene Isomerization | 8.0 – 12.0 | p-Xylene Approach to Equilibrium > 95%, Low Dealkylation | Non-metallic modifiers (e.g., Si) | 2024 |
Table 2: Characterization Techniques for B/L Ratio Assessment
| Technique | Acidity Probe | Information Gained | Limitations |
|---|---|---|---|
| Pyridine-adsorbed IR (Py-IR) | Pyridine | Quantifies Brønsted (1545 cm⁻¹) & Lewis (1450 cm⁻¹) sites; Acid strength distribution | Requires vacuum, semi-quantitative |
| NH₃-Temperature Programmed Desorption (NH₃-TPD) | Ammonia | Total acid amount, strength distribution | Cannot distinguish B vs. L sites alone |
| ²⁷Al MAS NMR | N/A | Identifies framework (Brønsted) vs. extra-framework (Lewis) Al | Quantitative but requires calibration |
| Iso-propylamine Decomposition (IPAD) | Iso-propylamine | Selective titration of Brønsted sites | Indirect calculation of Lewis sites |
Experimental Protocols
Protocol for Tailoring B/L Ratio in H-ZSM-5
Objective: Synthesize H-ZSM-5 samples with a targeted B/L ratio via post-synthetic modification. Materials: Parent H-ZSM-5 (SiO₂/Al₂O₃ = 30-80), Ammonium nitrate (NH₄NO₃), Metal precursor salts (e.g., Mg(NO₃)₂), Tetraethyl orthosilicate (TEOS), (NH₄)₂HPO₄, Muffle furnace, Tubular reactor for steaming. Procedure:
- Ion Exchange: Convert Na-ZSM-5 to NH₄-ZSM-5 via 1M NH₄NO₃ solution at 80°C for 2h (repeat 3x). Calcinate at 550°C for 5h to obtain H-ZSM-5 (Baseline).
- Dealumination (Increase Si/Al, modifies B/L): For controlled dealumination, treat H-ZSM-5 with 0.1-0.5M citric acid at 75°C for 4h. Wash, dry, calcine. Creates mesoporosity and extra-framework Al (Lewis sites).
- Metal Impregnation (Introduces Lewis Sites): Use incipient wetness impregnation with Mg(NO₃)₂ solution. Dry at 110°C overnight, calcine at 550°C for 4h. Mg²⁺ cations neutralize framework Al-OH, converting Brønsted to Lewis sites.
- Silanation / Phosphorylation (Passivates Lewis Sites): Vapor-phase or liquid-phase deposition of TEOS or (NH₄)₂HPO₄, followed by calcination. Selectively covers strong Lewis sites or reacts with framework Al.
- Steaming (Controlled Framework Dealumination): Treat H-ZSM-5 in a flow of 20-100% steam/N₂ at 500-700°C for 1-6h. Creates extra-framework Al clusters (Lewis sites) and reduces Brønsted density.
Protocol for Catalytic Evaluation in MTO
Objective: Test catalyst performance (activity, selectivity, lifetime) in a fixed-bed reactor. Setup: Stainless-steel tubular reactor (ID: 8 mm), online GC with FID, methanol feed pump, temperature-controlled oven. Procedure:
- Load 0.5 g catalyst (40-60 mesh) diluted with SiC. Pre-treat in N₂ at 550°C for 1h.
- Set reactor temperature to 400-480°C. Feed methanol at WHSV = 1-4 h⁻¹.
- Product analysis performed online every 30 min. Key metrics: Methanol conversion, selectivity to ethylene (C₂=), propylene (C₃=), and C₄⁺ olefins. Lifetime defined as time to 90% methanol conversion.
- Spent catalyst analyzed by TGA for coke content.
Key Diagrams
Diagram Title: Influence of B/L Ratio on MTO Reaction Pathways
Diagram Title: B/L Ratio Tuning via Post-Synthetic Modifications
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for B/L Ratio Research
| Item / Reagent | Primary Function in Research | Technical Note |
|---|---|---|
| H-ZSM-5 Zeolite (Reference) | Baseline catalyst with known SiO₂/Al₂O₃ ratio (e.g., 30, 50, 80). | Source from certified suppliers (e.g., Zeolyst, ACS Material) for reproducibility. |
| Pyridine (Anhydrous, >99.9%) | Probe molecule for in-situ or ex-situ FTIR to quantify B & L sites. | Must be thoroughly dried and stored under inert atmosphere. Toxic, handle in fume hood. |
| Tetraethyl Orthosilicate (TEOS) | Silanizing agent for selective passivation of external surface and Lewis acid sites. | Vapor-phase deposition yields more uniform coverage than liquid-phase. |
| Ammonium Metatungstate / Molybdate | Precursors for hydrotreating (W, Mo) metals in hydrocracking catalyst studies. | |
| Magnesium Nitrate Hexahydrate | Common source of Mg²⁺ for ion exchange to selectively convert Brønsted to Lewis sites. | Calcine post-impregnation at 500°C to decompose to MgO species. |
| NH₄NO₃ for Ion Exchange | Converts commercial Na-ZSM-5 to the active H-form (via NH₄-form). | Multiple exchanges required to achieve >95% H⁺ exchange. |
| Citric Acid / EDTA | Mild chelating agents for controlled framework dealumination. | Creates hierarchical porosity while generating extra-framework Al (Lewis sites). |
| n-Hexane / Iso-propylamine | Probe molecules for micropore volume assessment and Brønsted site titration (IPAD), respectively. | IPAD coupled with TPD-MS is a quantitative method for Brønsted acidity. |
Overcoming Challenges: Troubleshooting Deactivation and Optimizing B/L Ratio for Stability
Within the broader thesis on Brønsted to Lewis (B/L) acid site ratio research in H-ZSM-5 zeolites, this technical guide details the common analytical pitfalls leading to inaccurate acid site quantification and the unintended manipulation of the B/L ratio during sample handling and analysis. Accurate determination of this ratio is critical for rational catalyst design in petrochemical and pharmaceutical intermediate synthesis.
H-ZSM-5 is a cornerstone solid acid catalyst. Its catalytic activity and selectivity are governed not just by total acidity but by the precise ratio of proton-donating Brønsted (B) sites to electron-accepting Lewis (L) sites. Inaccurate characterization or unintended ratio shifts during experimental protocols lead to irreproducible results and flawed structure-activity correlations, jeopardizing downstream applications, including in drug precursor synthesis.
Pitfall 1: Inaccurate Characterization
Common techniques like Fourier-Transform Infrared Spectroscopy (FTIR) with probe molecules (e.g., pyridine, NH₃-Temperature Programmed Desorption (TPD)), and Solid-State Nuclear Magnetic Resonance (SSNMR) are prone to methodological errors.
Quantitative Data from Common Characterization Techniques
Table 1: Comparison of Acid Site Quantification Techniques and Associated Pitfalls
| Technique | Probe Molecule | Typical B/L Range Reported | Key Pitfall | Impact on Ratio |
|---|---|---|---|---|
| FTIR-Pyridine | Pyridine | 0.5 - 5.0 | Inadequate degassing, leading to physisorbed pyridine; incorrect extinction coefficients. | Over/under-estimation of both sites; ratio skewed. |
| NH₃-TPD | Ammonia | 1.0 - 10.0 | Overlapping desorption peaks; diffusion limitations; decomposition. | Misassignment of peak areas; inaccurate ratio. |
| ¹H SSNMR | None (direct) | 2.0 - ∞ | Low sensitivity; interference from silanol groups; quadrupolar interactions. | Underestimation of Lewis sites; ratio inflated. |
| XPS | None (surface) | Varies widely | Extreme surface sensitivity (<10 nm); charging effects; complex deconvolution. | Not representative of bulk; ratio misleading. |
Detailed Experimental Protocol: FTIR-Pyridine withIn SituCell
Objective: To accurately quantify Brønsted and Lewis acid sites in H-ZSM-5. Materials: H-ZSM-5 wafer (5-10 mg/cm²), in situ quartz IR cell with heating, high-vacuum system (<10⁻⁵ mbar), pyridine vapor source. Procedure:
- Pre-treatment: Place wafer in cell. Heat to 450°C under dynamic vacuum (1-2 hours) to remove adsorbates.
- Background Scan: Cool to 150°C. Acquire background IR spectrum.
- Adsorption: Expose to saturated pyridine vapor at 150°C for 15 min.
- Desorption: Evacuate at 150°C for 30 min to remove physisorbed pyridine.
- Measurement: Record spectrum at 150°C.
- Quantification: Integrate bands at ~1545 cm⁻¹ (B site, pyridinium ion) and ~1455 cm⁻¹ (L site, coordinated pyridine). Use published extinction coefficients (e.g., E(B) ≈ 1.67 cm/μmol, E(L) ≈ 2.22 cm/μmol) to calculate site densities.
Pitfall Avoidance: The evacuation step at 150°C is critical. Omitting it inflates both B and L intensities, but not necessarily proportionally, altering the reported B/L ratio.
Title: FTIR-Pyridine Workflow with Critical Evacuation Step
Pitfall 2: Unintended Ratio Shifts
The B/L ratio is not a fixed material property but can be altered by common laboratory procedures.
Mechanisms of Unintended Changes
- Dealumination: Steam or high-temperature calcination can extract aluminum from the framework, converting a Brønsted site (bridging Si-OH-Al) into an extra-framework Al Lewis site, decreasing the B/L ratio.
- Coking: During catalytic testing, carbonaceous deposits preferentially block Brønsted sites, artificially lowering the apparent B ratio.
- Incomplete Activation: Failure to fully convert the NH₄⁺-form to the H⁺-form (Brønsted site) leaves inactive material, underestimating B acidity.
Experimental Protocol: NH₄-ZSM-5 to H-ZSM-5 Activation
Objective: To fully activate Brønsted sites without causing dealumination. Materials: NH₄-ZSM-5 powder, tube furnace, controlled dry air or inert gas flow. Procedure:
- Place powder in a shallow bed within a quartz boat.
- Insert into furnace with a gas flow of 50 ml/min (dry air or N₂).
- Heat with a slow ramp rate (2°C/min) to 550°C.
- Hold at 550°C for 5 hours.
- Cool under dry gas flow to room temperature.
- Transfer immediately to an inert atmosphere or sealed container.
Pitfall Avoidance: A fast ramp rate or temperatures exceeding 600°C can induce framework dealumination, creating extra-framework Al (Lewis sites) and permanently reducing the intended B/L ratio.
Title: Activation Protocol Impact on Final B/L Ratio
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 2: Key Reagents and Materials for H-ZSM-5 B/L Ratio Research
| Item | Function & Rationale | Critical Specification |
|---|---|---|
| H-/NH₄-ZSM-5 (Reference) | Benchmark material with certified SiO₂/Al₂O₃ ratio. | Known provenance (e.g., Zeolyst, Clariant). SiO₂/Al₂O₃ = 40 recommended for clear spectral features. |
| Pyridine, Spectroscopy Grade | Probe molecule for FTIR distinction of B vs. L sites. | Anhydrous (water-free), ≥99.9% purity. Stored over molecular sieve. |
| Ammonia, 5% in He (for TPD) | Probe for total acid strength distribution. | High-purity gas mix. Moisture and impurity levels < 10 ppm. |
| In Situ IR Cell/DRIFTS | Allows sample pre-treatment and probe adsorption under controlled conditions. | With temperature controller, vacuum/gas manifold, and KBr windows. |
| Quartz Wool/Tubes | For sample packing in TPD/MS experiments. | Acid-washed and pre-calcined at 700°C to remove organics. |
| Deuterated Acetonitrile (CD₃CN) | Alternative probe for FTIR/SSNMR; different steric and electronic properties vs. pyridine. | D-content >99.8%. |
| Magic Angle Spinning (MAS) NMR Rotors | For SSNMR analysis of acid sites and framework integrity. | Zirconia rotors (e.g., 4 mm OD). |
Integrated Workflow for Reliable B/L Assessment
A multi-technique approach is necessary to cross-validate results and diagnose pitfalls.
Title: Multi-Technique Validation Pathway for B/L Ratio
Accurate characterization of the Brønsted to Lewis acid site ratio in H-ZSM-5 requires strict adherence to validated protocols and an awareness of how common laboratory practices can unintentionally alter this critical parameter. By implementing the detailed methodologies and validation workflows outlined herein, researchers can generate robust, reproducible data essential for advancing catalyst development in both petrochemical and pharmaceutical synthesis fields.
Within the broader thesis on Brønsted (B) to Lewis (L) acid site ratio in H-ZSM-5, this whitepaper investigates a critical performance determinant: catalyst deactivation via coking. The B/L ratio is not merely a descriptor of acid site distribution; it is a governing factor in reaction pathways, dictating the nature, rate, and selectivity of coke formation. This guide details the mechanistic interplay between acid site ratio and coke-induced deactivation, providing technical protocols and data for researchers in catalysis and process development.
Coke formation in H-ZSM-5 proceeds via sequential reactions of hydrocarbon intermediates. The B/L ratio steers these reactions:
- Brønsted Acid Sites (BAS): Catalyze protolytic cracking, oligomerization, cyclization, and hydride transfer reactions, leading to aromatic coke precursors (e.g., alkylbenzenes, naphthalenes).
- Lewis Acid Sites (LAS): Facilitate dehydrogenation and polymerization reactions, promoting the formation of polyaromatic and graphitic-like coke species. LAS can also arise from framework dealumination, often accelerated by coke formation itself.
A high B/L ratio favors "softer," internally confined aromatic coke, while a low B/L (or high LAS concentration) promotes "harder," external polycyclic aromatic coke that blocks pore mouths. The dynamic conversion of BAS to LAS during reaction further complicates the deactivation trajectory.
Experimental Protocols for Investigating B/L Ratio Effects
3.1. Catalyst Preparation & Characterization Protocol
- Materials: NH₄-ZSM-5 (target Si/Al), deionized water, calcination furnace.
- Procedure:
- Ion Exchange: Convert commercial NH₄-ZSM-5 to H-ZSM-5 via calcination in static air at 550°C for 5 hours.
- Post-Synthetic Modification: To vary B/L ratio:
- Steaming: Treat H-ZSM-5 with 20-100% steam at 500-700°C to selectively remove framework Al, creating non-framework Al (LAS).
- Acid Leaching: Wash steamed samples with mild nitric acid to remove extra-framework Al, partially restoring a high B/L ratio.
- Characterization:
- Acid Site Quantification: Perform Ammonia Temperature-Programmed Desorption (NH₃-TPD) to measure total acidity. Use Pyridine-adsorbed Fourier-Transform Infrared Spectroscopy (Py-FTIR) at 150°C (for BAS & LAS) and 450°C (for strong sites) to calculate the precise B/L ratio from integrated band areas (1545 cm⁻¹ for BAS, 1450 cm⁻¹ for LAS).
- Textural Properties: Conduct N₂ physisorption to determine BET surface area and micropore volume.
3.2. Catalytic Testing & Coke Analysis Protocol
- Reaction: Model reaction: n-hexane cracking or methanol-to-hydrocarbons (MTH) in a fixed-bed tubular reactor.
- Conditions: T=350-450°C, P=1 atm, WHSV= 1-10 h⁻¹. Time-on-stream (TOS) experiments run for 2-24 hours.
- Procedure:
- Catalysts are activated in-situ at 450°C under N₂.
- Reactant feed is initiated, and effluent products are analyzed periodically by online GC.
- After reaction, the reactor is cooled under N₂. Spent catalyst is divided for analysis.
- Coke Characterization:
- Thermogravimetric Analysis (TGA): Burn coke in air (ramp to 800°C) to determine total coke amount and oxidation temperature (indicator of coke hardness).
- Solvent Extraction & GC-MS: Dissolve soluble coke species (alkyl aromatics) in dichloromethane and analyze.
- ¹³C Solid-State NMR: Identify the nature of insoluble, polyaromatic coke.
Data Presentation: Quantitative Impact of B/L Ratio
Table 1: Catalyst Properties and Coke Formation Data
| Catalyst ID | Si/Al | Treatment | B/L Ratio* (Py-IR, 150°C) | Total Coke (wt%, TGA) | Coke Burn-Off Peak Temp. (°C) | Relative Deactivation Rate† |
|---|---|---|---|---|---|---|
| ZSM-5-H | 25 | None | 4.2 | 8.5 | 525 | 1.0 (baseline) |
| ZSM-5-S | 25 | Steaming | 0.8 | 12.1 | 620 | 3.5 |
| ZSM-5-AL | 25 | Acid-Leach | 3.0 | 7.2 | 540 | 1.8 |
| ZSM-5-LA | 40 | None | 5.5 | 6.0 | 515 | 0.7 |
*B/L Ratio = Integrated area of Band at 1545 cm⁻¹ / 1450 cm⁻¹. †Defined as the inverse of time to reach 50% initial activity conversion in n-hexane cracking.
Table 2: Coke Selectivity as a Function of B/L Ratio (GC-MS of Extracted Coke)
| Coke Species Class | Example Compounds | ZSM-5-H (B/L=4.2) | ZSM-5-S (B/L=0.8) |
|---|---|---|---|
| Mono-/Di-alkylbenzenes | Toluene, Xylenes | High | Low |
| Polyalkylbenzenes (C10+) | Pentamethylbenzene | Moderate | Very Low |
| Indanes & Tetralins | Methylindane | Moderate | Low |
| Naphthalenes | Methylnaphthalene | Low | High |
| Anthracenes/Phenanthrenes | - | Trace | Moderate |
The Scientist's Toolkit: Key Research Reagent Solutions
| Item / Reagent | Function / Explanation |
|---|---|
| NH₄-ZSM-5 Zeolites | Starting material with varying Si/Al ratios to define initial acid site density. |
| Pyridine (anhydrous) | Probe molecule for FTIR to quantify and distinguish Brønsted vs. Lewis acid sites. |
| n-Hexane / Methanol | Model reactants for acid-catalyzed cracking (n-hexane) or aromatization (Methanol-to-Hydrocarbons, MTH) to study coke formation under controlled conditions. |
| Steam Generator | Precise equipment to create partial pressure of water for controlled hydrothermal dealumination to adjust B/L ratio. |
| Nitric Acid (0.1-1M) | For post-steam acid leaching to remove extra-framework Al without extensive framework damage. |
| Dichloromethane (DCM) | Solvent for extracting soluble (precursor) coke species from spent catalysts for GC-MS analysis. |
| Calibration Gas Mixtures (H₂, He, Air) | Essential for GC-TCD/FID calibration and TGA/MS gas analysis during coke oxidation. |
Visualization of Pathways and Workflows
Diagram 1: Coke Formation Pathways Dictated by Acid Sites
Diagram 2: Experimental Workflow for B/L-Coke Study
This whitepaper is framed within a broader thesis research on the precise comparison and control of Brønsted (BAS) to Lewis (LAS) acid site ratios in H-ZSM-5 zeolites. The catalytic performance in numerous reactions, from hydrocarbon cracking to drug intermediate synthesis, is critically dependent on this ratio. However, under severe operational conditions (high temperature, steam), dealumination—the removal of framework aluminum—is inevitable. This process irreversibly alters the targeted BAS/LAS ratio, converting strong Brønsted sites into extra-framework aluminum (EFAL) Lewis sites and eventually leading to mesopore formation and complete deactivation. Mitigating dealumination is therefore paramount to preserving catalyst integrity and function for industrial applications.
Core Mechanisms of Dealumination in H-ZSM-5
Steam-mediated hydrolysis is the primary pathway: [ \text{Si-O-Al(OH)-Si} + H2O \rightarrow \text{Si-OH} + \text{HO-Al-Si} \rightarrow \text{Si-OH} + \text{Si-OH} + \text{Al(OH)}3 \text{(EFAL)} ] The resulting EFAL species can occupy channel intersections, block active sites, and introduce uncontrolled Lewis acidity. The rate is influenced by temperature, steam partial pressure, framework aluminum density, and crystal defects.
Table 1: Impact of Severe Conditions (750°C, 100% Steam) on H-ZSM-5 Acidic Properties
| Zeolite Sample (Si/Al=15) | Treatment Duration (hr) | BAS (µmol/g) | LAS (µmol/g) | BAS/LAS Ratio | Relative Crystallinity (%) |
|---|---|---|---|---|---|
| Parent H-ZSM-5 | 0 | 450 | 120 | 3.75 | 100 |
| After Dealumination | 5 | 210 | 310 | 0.68 | 85 |
| After Dealumination | 20 | 95 | 280 | 0.34 | 72 |
Table 2: Efficacy of Stabilization Strategies on BAS/LAS Ratio Preservation
| Mitigation Strategy | Example Protocol | Post-Treatment BAS/LAS Ratio (vs. Parent) | Key Mechanism of Action |
|---|---|---|---|
| Steam Treatment (Mild) | 550°C, 20% H2O, 2h | 3.1 (83% retained) | Pre-emptive removal of defect Al, healing |
| Phosphorus Promotion | 1.0 wt% H3PO4 impregnation | 3.4 (91% retained) | Al-O-P bonding, diffusion barrier formation |
| Lanthanum Exchange | 0.5 wt% La via ion exchange | 3.6 (96% retained) | Electrostatic stabilization of framework Al |
| Silanation | Chem. vapor dep. of TEOS | 2.9 (77% retained) | External surface passivation, pore mouth narrowing |
| Combinatorial (P/La) | 0.8% P + 0.3% La | 3.7 (99% retained) | Synergistic stabilization |
Experimental Protocols for Key Mitigation Strategies
Protocol 4.1: Pre-Steam Stabilization (Mild Hydrothermal Treatment)
- Material: Pelletized H-ZSM-5 (Si/Al = 15-40), sieved to 180-250 µm.
- Procedure: Load 2g of zeolite into a fixed-bed quartz reactor.
- Conditioning: Purge with dry N2 (50 ml/min), heat to 550°C at 5°C/min.
- Treatment: Switch to a gas stream saturated with H2O at 70°C (≈20% steam in N2). Maintain for 2-6 hours.
- Cooling: Switch back to dry N2, cool to room temperature.
- Analysis: Measure BAS/LAS via pyridine FTIR (150°C desorption). Calculate crystallinity via XRD.
Protocol 4.2: Phosphorus Promotion via Wet Impregnation
- Solution Prep: Dissolve di-ammonium hydrogen phosphate ((NH4)2HPO4) in deionized water to achieve 0.5-2.0 wt% P relative to zeolite.
- Impregnation: Add the solution dropwise to 5g of H-ZSM-5 under continuous stirring. Use incipient wetness volume.
- Aging: Let the mixture stand for 2 hours at room temperature.
- Drying: Dry at 110°C for 12 hours in a static oven.
- Calcination: Heat in muffle furnace in air to 550°C (2°C/min ramp) and hold for 5 hours. This converts phosphorus to its oxide form, which migrates and reacts with framework Al.
Protocol 4.3: Acid Site Characterization via Pyridine FTIR
- Pellet Prep: Press 15-20 mg of zeolite into a self-supporting wafer under 5 tons.
- Pre-Treatment: Place wafer in a high-temperature IR cell, heat under vacuum (<10^-3 Pa) to 450°C for 2h to desorb water.
- Cool & Adsorb: Cool to 150°C, expose to pyridine vapor (≈5 mbar) for 15 min.
- Desorption: Evacuate at 150°C for 30 min to remove physisorbed pyridine.
- Spectrum Acquisition: Record IR spectrum between 1400-1600 cm^-1.
- Quantification: Use molar extinction coefficients (ε) to calculate concentrations:
- BAS: Band at ~1545 cm^-1 (ε = 0.073 cm/µmol)
- LAS: Band at ~1455 cm^-1 (ε = 0.101 cm/µmol)
- [ C = \frac{A \cdot S}{ε \cdot m} ] where A=absorbance, S=wafer area (cm^2), m=sample mass (g).
Visualization of Strategies and Pathways
Diagram 1: Dealumination Pathways vs. Mitigation Strategies (78 chars)
Diagram 2: Experimental Workflow for Stability Testing (58 chars)
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for Acid Site Ratio Research
| Item Name & Specification | Function/Relevance | Brief Explanation of Role |
|---|---|---|
| NH4-ZSM-5 (Zeolyst Intl.) | Parent Catalyst | Provides the framework material for proton exchange (H-form) and subsequent modification. Consistent Si/Al ratio is critical. |
| Pyridine, 99.9+%, anhydrous (Sigma-Aldrich) | BAS/LAS Probe Molecule | Selective adsorbate for infrared spectroscopy. Pyridine coordinates to Lewis sites and protonates at Brønsted sites. |
| Di-ammonium hydrogen phosphate, (NH4)2HPO4 | Phosphorus Source | Common precursor for phosphorus promotion. Decomposes on calcination to PxOy species that bind to framework Al. |
| Lanthanum(III) nitrate hexahydrate | Lanthanum Source | Used in aqueous ion exchange to introduce La3+ cations that electrostatically stabilize framework Al atoms. |
| Tetraethyl orthosilicate (TEOS) | Silanation Agent | Volatile silicon source for chemical vapor deposition to passivate external surfaces and modify pore mouth size. |
| Deuterated Acetonitrile (CD3CN) | Alternative Probe Molecule | Used in solid-state NMR for acid site characterization, with different 13C chemical shifts for BAS vs. LAS. |
| In-situ IR Cell (HTC, Harrick) | Characterization Tool | Allows for controlled temperature, gas environment, and adsorption/desorption cycles during FTIR measurement. |
This technical guide is framed within the context of a broader thesis investigating the strategic manipulation of Brønsted (B) to Lewis (L) acid site ratios in H-ZSM-5 zeolites. The central premise is that catalytic lifetime—a critical metric for industrial viability—is not solely a function of total acidity but is intricately governed by the equilibrium between active site density (driving activity) and the density of sites that promote coke formation and deactivation (impacting stability). By moving beyond the simplistic maximization of Brønsted sites, we explore how controlled synthesis and post-synthetic modifications can engineer an optimal B/L ratio, thereby achieving a catalyst that balances high initial activity with prolonged operational stability.
Fundamentals of Acid Sites in H-ZSM-5
H-ZSM-5 possesses a framework of bridged hydroxyl groups (Si–OH–Al), which are the prototypical Brønsted acid sites. Lewis acid sites are typically coordinatively unsaturated Al species, which can originate from framework defects or extra-framework aluminum (EFAL). The interplay between these sites dictates catalytic behavior:
- Brønsted Sites (B): Provide proton-donating capability, essential for reactions like cracking, isomerization, and alkylation.
- Lewis Sites (L): Act as electron-pair acceptors. They can modify reaction pathways, often synergizing with B sites (creating B-L pairs), but can also catalyze undesirable condensation reactions leading to coke.
Synthesis and Modification Protocols for B/L Ratio Control
Protocol 3.1: Direct Hydrothermal Synthesis for Framework Al Density Control
- Objective: Synthesize H-ZSM-5 with varying bulk Si/Al ratios to control total framework Al (primary source of Brønsted sites).
- Methodology:
- Prepare a gel with molar composition:
x SiO₂ : 0.1x Al₂O₃ : 0.4 TPAOH : 30 H₂O, wherexis varied (e.g., 30, 60, 100) to target Si/Al ratios of 15, 30, and 50. - Stir the gel at room temperature for 24 hours.
- Transfer to a Teflon-lined stainless-steel autoclave. Crystallize at 170°C for 48 hours under autogenous pressure.
- Recover by filtration, wash with deionized water, and dry at 100°C overnight.
- Calcine in static air at 550°C for 6 hours (heating rate: 2°C/min) to remove the template.
- Convert to H-form via triple ion exchange with 1 M NH₄NO₃ solution (80°C, 2 hours each), followed by drying and calcination at 500°C for 4 hours.
- Prepare a gel with molar composition:
Protocol 3.2: Post-Synthetic Dealumination to Generate Lewis Sites
- Objective: Create controlled amounts of extra-framework Al (EFAL) Lewis sites via steam dealumination.
- Methodology:
- Place 2g of parent H-ZSM-5 (Si/Al=30) in a quartz tube reactor.
- Under a flow of 20 vol% H₂O/N₂ (total flow 100 mL/min), treat the sample at 500-700°C (e.g., 550°C, 600°C, 650°C) for 2 hours.
- Cool to room temperature under dry N₂ flow. This process hydrolyzes framework Al, generating EFAL species while reducing Brønsted site density.
Protocol 3.3: Mild Acid Washing for Selective Lewis Site Removal
- Objective: Remove extra-framework Al (EFAL) Lewis sites while preserving framework Brønsted sites.
- Methodology:
- Disperse 2g of steam-dealuminated zeolite in 100 mL of 0.1 M HNO₃ solution.
- Stir the suspension at 80°C for 3 hours.
- Filter, wash thoroughly with deionized water until filtrate pH is neutral.
- Dry at 100°C overnight and calcine at 450°C for 2 hours.
Characterization and Quantitative Data
Table 1: Acid Site Properties of Modified H-ZSM-5 Catalysts Data derived from NH₃-TPD and Pyridine FTIR. B/L ratio calculated from IR band areas at 1545 cm⁻¹ (B) and 1450 cm⁻¹ (L).
| Sample ID | Synthesis Si/Al | Treatment | Total Acidity (mmol NH₃/g) | Brønsted (μmol/g) | Lewis (μmol/g) | B/L Ratio |
|---|---|---|---|---|---|---|
| Z-15 | 15 | Parent (H-form) | 0.89 | 420 | 35 | 12.0 |
| Z-30 | 30 | Parent (H-form) | 0.61 | 310 | 28 | 11.1 |
| Z-30-S600 | 30 | Steam, 600°C, 2h | 0.53 | 205 | 112 | 1.83 |
| Z-30-S600-AW | 30 | Steam 600°C + Acid Wash | 0.48 | 238 | 45 | 5.29 |
Table 2: Catalytic Performance in Methanol-to-Hydrocarbons (MTH) at 370°C Lifetime defined as time to 50% methanol conversion. Initial activity measured at TOS=10 min.
| Sample ID | B/L Ratio | Initial Activity (molCH₃OH/g·h) | Coke at Deactivation (wt%) | Lifetime (h) |
|---|---|---|---|---|
| Z-15 | 12.0 | 4.2 | 18.5 | 48 |
| Z-30 | 11.1 | 3.8 | 15.1 | 65 |
| Z-30-S600 | 1.83 | 2.5 | 22.7 | 28 |
| Z-30-S600-AW | 5.29 | 3.3 | 16.8 | 72 |
Mechanistic Pathways and Deactivation
The B/L ratio directly influences the hydrocarbon pool mechanism and coke formation pathways in acid-catalyzed reactions like MTH.
Figure 1: Acid Site Roles in Reaction & Coke Pathways
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Acid Site Density Research
| Item / Reagent | Function / Purpose | Key Consideration |
|---|---|---|
| Tetrapropylammonium hydroxide (TPAOH) 40% | Structure-directing agent (SDA) for ZSM-5 synthesis. | High purity to avoid alkali metal contamination. |
| Fumed Silica (SiO₂) | Silicon source for hydrothermal synthesis. | High surface area ensures complete dissolution in gel. |
| Sodium Aluminate (NaAlO₂) | Aluminum source for framework incorporation. | Exact Al content must be known for stoichiometry. |
| Ammonium Nitrate (NH₄NO₃) | For ion exchange to convert Na-ZSM-5 to H-ZSM-5. | Multiple exchanges required for complete H⁺ form. |
| Pyridine, 99.8% (anhydrous) | Probe molecule for FTIR distinction of B vs. L sites. | Must be rigorously dried and handled under inert atmosphere. |
| Nitric Acid (HNO₃), 0.1M Solution | For selective leaching of extra-framework Al (EFAL). | Mild concentration and temperature prevent framework damage. |
| Steam Generation Setup | For controlled steam dealumination treatments. | Requires precise temperature and H₂O partial pressure control. |
| NH₃ for TPD | Probe for total acidity measurement via Temperature-Programmed Desorption. | Use ultra-high purity gas with an inline moisture trap. |
Experimental Workflow for Lifetime Assessment
Figure 2: Workflow for Catalyst Design & Testing
The optimization of catalytic lifetime necessitates a deliberate departure from merely maximizing acid site density. The data presented confirm that an intermediate B/L ratio (achieved, for example, via controlled dealumination followed by mild acid wash) often yields the optimal balance between sufficient activity and enhanced stability. The strategic generation and subsequent management of Lewis acid sites are paramount. Future research directions should focus on advanced spectroscopic and computational methods to precisely map the spatial distribution and synergistic coupling of B and L sites, enabling the next generation of designed-for-lifetime zeolite catalysts.
1. Introduction: The Critical Role of B/L Ratio in Catalytic Performance
Within the broader thesis on Brønsted (B) to Lewis (L) acid site ratio comparisons in H-ZSM-5, catalyst deactivation represents a pivotal challenge. Spent H-ZSM-5 catalysts, typically deactivated by coke deposition and framework dealumination, suffer from a disrupted B/L acid site ratio. This ratio is fundamental for directing reaction pathways in processes like methanol-to-hydrocarbons (MTH), fluid catalytic cracking (FCC), and biomass valorization. Brønsted acid sites (bridging hydroxyl groups) are primary active sites for many acid-catalyzed reactions, while Lewis acid sites (extra-framework aluminum, EFAL) can modify selectivity and stability. An optimal B/L ratio ensures both high activity and desired product distribution. This whitepaper details targeted regeneration protocols designed not merely to remove coke but to strategically restore the native B/L ratio, thereby recovering the intrinsic catalytic fingerprint.
2. Quantitative Data on Deactivation and Regeneration Outcomes
Table 1: Common Changes in Acidic Properties of H-ZSM-5 Upon Deactivation
| Property | Fresh H-ZSM-5 | Spent H-ZSM-5 (Coked/Dealuminated) | Primary Cause of Change |
|---|---|---|---|
| Total Acidity (mmol NH₃/g) | 0.4 - 0.8 | 0.1 - 0.3 | Pore blocking by coke, site coverage. |
| Brønsted Acid Sites (μmol/g) | 300 - 600 | 50 - 200 | Direct coverage by polyaromatic coke; framework dealumination. |
| Lewis Acid Sites (μmol/g) | 50 - 150 | 80 - 250 | Increase due to formation of extra-framework Al (EFAL) from steam. |
| B/L Ratio | 4 - 10 | 0.5 - 2.5 | Severe decrease due to B-site loss and L-site gain. |
| Micropore Volume (cm³/g) | 0.15 - 0.18 | 0.05 - 0.10 | Occupied by coke deposits. |
Table 2: Efficacy of Different Regeneration Protocols in Restoring B/L Ratio
| Protocol | Key Steps | Impact on Coke | Impact on Brønsted Sites | Impact on Lewis Sites | Restored B/L Ratio (Typical Range) |
|---|---|---|---|---|---|
| Simple Calcination | Air, 550°C, 4-8 h. | Effective removal (>95%). | Cannot restore dealuminated sites; may create new EFAL. | Can increase EFAL from harsh oxidation. | Low (1.5 - 3.5) |
| Steam Treatment + Calcination | Mild steam (e.g., 10% H₂O), 500-600°C, then calcination. | Removes coke; can hydrothermally dealuminate. | Significant further dealumination, permanent loss. | Large increase in EFAL species. | Very Low (0.5 - 2.0) |
| Organosilane Treatment | e.g., Tetramethoxysilane vapor deposition post-calcination. | N/A (post-coke removal). | Does not directly restore. | Selective poisoning/passivation of strong Lewis sites. | Artificially High (5 - 12) by masking L. |
| Combined Acid Leaching & Mild Calcination | 1. Mild Calcination (450°C). 2. Controlled HNO₃ or Oxalic Acid Leach. 3. Final Mild Calcination. | Removed in step 1. | Partial restoration via removal of EFAL that block B sites. | Selective removal of non-framework Lewis Al. | Optimized Restoration (3.5 - 7.0) |
3. Detailed Experimental Protocols
Protocol A: Standard Oxidative Calcination for Coke Removal
- Setup: Place 1.0 g of spent H-ZSM-5 catalyst in a quartz boat within a horizontal tube furnace.
- Gas Flow: Establish a dry air flow (≥ 99.9% purity) at 50 mL/min.
- Temperature Program: Heat from room temperature to 550°C at a ramp rate of 2°C/min.
- Hold: Maintain at 550°C for 6 hours under continuous air flow.
- Cool Down: Purge with inert gas (N₂) while cooling to room temperature.
- Analysis: Weigh to determine coke burn-off. Characterize acidity via NH₃-TPD or pyridine FTIR.
Protocol B: Combined Acid Leaching for B/L Ratio Restoration (Recommended) Objective: To remove coke and selectively etch extra-framework aluminum (EFAL) to unblock and reveal framework Brønsted sites.
- Mild Coke Burn-Off: Perform Protocol A, but with a final temperature of 450°C for 4 hours to minimize hydrothermal damage.
- Acid Leaching: Transfer the calcined catalyst to a round-bottom flask.
- Leaching Solution: Add 100 mL of 0.2 M oxalic acid (or 0.1 M HNO₃) solution per gram of catalyst.
- Reaction: Reflux the mixture at 100°C for 2 hours with constant stirring.
- Washing: Filter the solid catalyst and wash extensively with deionized water (until filtrate pH is neutral).
- Drying: Dry the catalyst at 120°C overnight.
- Final Activation: Perform a second, mild calcination in dry air at 450°C for 2 hours to remove residual organics and re-form the acid site structure.
4. Visualization of Protocols and Acid Site Evolution
Diagram Title: Regeneration Pathways & Acid Site Fate in H-ZSM-5
Diagram Title: Combined Acid Leaching Regeneration Protocol Workflow
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for B/L Ratio Restoration Studies
| Reagent / Material | Specification / Grade | Primary Function in Protocol |
|---|---|---|
| Spent H-ZSM-5 Catalyst | Si/Al ratio 15-40, defined particle size. | The substrate for regeneration; baseline for B/L ratio restoration studies. |
| Dry Air Supply | ≥ 99.9% purity, moisture trap equipped. | Oxidizing atmosphere for controlled coke combustion during calcination. |
| Oxalic Acid Dihydrate | Analytical Reagent (AR) Grade, >99%. | Chelating agent for selective dissolution of extra-framework aluminum (EFAL) species. |
| Nitric Acid (HNO₃) | 65-70% w/w, TraceMetal Grade. | Alternative strong inorganic acid for EFAL removal, less selective than oxalic acid. |
| Ammonium Nitrate (NH₄NO₃) | AR Grade, >99%. | For post-regeneration ion-exchange to convert any Na⁺-form back to H⁺-form if needed. |
| Pyridine | Anhydrous, 99.8%, dried over molecular sieve. | Probe molecule for FTIR spectroscopy to quantitatively distinguish Brønsted vs. Lewis acid sites. |
| Ammonia (NH₃) | 5% in He/Ar mix, for TPD. | Probe molecule for Temperature-Programmed Desorption (TPD) to measure total acidity and strength distribution. |
| Inert Gas (N₂ or Ar) | High Purity (≥ 99.999%). | For purging and cooling steps to prevent unintended re-adsorption or oxidation. |
Benchmarking Performance: Comparative Analysis of Catalysts with Engineered vs. Conventional B/L Ratios
1. Introduction This guide details the methodology for head-to-head validation of zeolite catalysts, specifically within the context of ongoing research into the Brønsted (B) to Lewis (L) acid site ratio in H-ZSM-5. The precise quantification and comparison of this ratio is critical for understanding catalytic performance in industrially relevant model reactions, such as the cracking of alkanes or the conversion of methanol to hydrocarbons (MTH). This document provides a standardized framework for experimental comparison, data presentation, and analysis.
2. Core Experimental Protocols
2.1 Catalyst Preparation & Acid Site Characterization
- Materials: NH4-ZSM-5 (various Si/Al ratios), deionized water, ammonium nitrate, tubular furnace.
- Protocol for H-ZSM-5 Activation: Ion exchange NH4-ZSM-5 with 1M NH4NO3 solution (80°C, 2h, repeated 3x), wash, dry (110°C, 12h), calcine in static air (550°C, 5h, 2°C/min ramp) to produce the protonic form (H-ZSM-5).
- Protocol for Pyridine FTIR (Quantification of B/L Ratio):
- Press catalyst into a self-supporting wafer (~10 mg/cm²).
- Load into a controlled-environment IR cell with KBr windows.
- Activate in situ under vacuum (450°C, 1h) to remove adsorbed species.
- Cool to 150°C and record background spectrum.
- Expose to pyridine vapor (equilibrium pressure ~6 Torr) for 15 min.
- Physically desorb at 150°C under vacuum for 30 min to remove physisorbed pyridine.
- Record spectrum in the 1400-1600 cm⁻¹ region.
- Quantify Brønsted (B) acid sites (band at ~1545 cm⁻¹) and Lewis (L) acid sites (band at ~1455 cm⁻¹) using published molar extinction coefficients (e.g., εB = 1.67 cm/μmol, εL = 2.22 cm/μmol).
- Protocol for Temperature-Programmed Desorption of Ammonia (NH3-TPD):
- Load 100 mg of catalyst into a quartz U-tube reactor.
- Pre-treat in He flow (30 mL/min) at 550°C for 1h.
- Cool to 100°C and saturate with 5% NH3/He for 30 min.
- Flush with He at 100°C for 1h to remove physisorbed NH3.
- Heat in He flow (10°C/min) to 700°C while monitoring desorbed NH3 via TCD or MS.
2.2 Catalytic Testing: n-Hexane Cracking as a Model Reaction
- Reactor System: Fixed-bed, continuous-flow, quartz microreactor (ID: 6 mm).
- Standard Reaction Conditions: T = 500°C, P = 1 atm, WHSV = 2 h⁻¹, catalyst mass = 250 mg (diluted with SiC), feed = 5 mol% n-hexane in N2.
- Procedure:
- Load catalyst, activate in situ under N2 at 500°C for 1h.
- Initiate hydrocarbon feed using a calibrated syringe pump.
- Analyze effluent gas stream after 15 min time-on-stream (TOS) using an online GC equipped with a capillary column (e.g., HP-PONA) and FID.
- Key Metrics Calculated:
- Conversion (%): (n-hexanein - n-hexaneout) / n-hexanein × 100.
- Selectivity to Product i (%): (Moles of product i formed) / (Total moles of n-hexane converted) × 100.
- Rate Constant (k, s⁻¹): Calculated assuming first-order kinetics for a differential reactor.
3. Quantitative Data Presentation
Table 1: Acidic Properties of H-ZSM-5 Catalysts with Varying B/L Ratio
| Catalyst ID | Si/Al Ratio (Bulk) | Brønsted Acidity (μmol/g) | Lewis Acidity (μmol/g) | B/L Ratio | Total Acidity (μmol/g) | NH3-TPD Peak Max (°C) |
|---|---|---|---|---|---|---|
| HZSM5-A | 15 | 420 | 85 | 4.94 | 505 | 365 |
| HZSM5-B | 25 | 310 | 105 | 2.95 | 415 | 350 |
| HZSM5-C | 40 | 195 | 125 | 1.56 | 320 | 340 |
| HZSM5-D* | 15 | 380 | 210 | 1.81 | 590 | 380 |
Note: HZSM5-D prepared via mild steaming to intentionally decouple B/L ratio from Si/Al.
Table 2: Catalytic Performance in n-Hexane Cracking (500°C, WHSV=2 h⁻¹)
| Catalyst ID | n-Hexane Conv. (%) | App. Rate Constant, k (s⁻¹) | Selectivity at ~20% Conv. (%) | |||
|---|---|---|---|---|---|---|
| C1-C2 | C3-C4 | C5+ | Aromatics | |||
| HZSM5-A | 22.5 | 1.58 | 12.1 | 45.2 | 28.4 | 14.3 |
| HZSM5-B | 18.7 | 1.27 | 10.5 | 48.7 | 31.5 | 9.3 |
| HZSM5-C | 14.2 | 0.92 | 8.8 | 52.1 | 35.8 | 3.3 |
| HZSM5-D | 25.8 | 1.95 | 15.7 | 42.9 | 25.1 | 16.3 |
4. Visualization of Workflow & Relationship
Diagram Title: Workflow for B/L Ratio Impact Validation
5. The Scientist's Toolkit: Key Research Reagent Solutions & Materials
| Item | Function in H-ZSM-5 B/L Research |
|---|---|
| NH4-ZSM-5 Zeolites (Various Si/Al) | The parent material. Si/Al ratio is the primary (but not sole) determinant of initial Brønsted acid site density. |
| Ammonium Nitrate (NH4NO3) | Used for ion exchange to prepare the pure protonic form (H-ZSM-5) from the ammonium form. |
| Pyridine (Spectroscopic Grade) | Probe molecule for FTIR spectroscopy. Selectively binds to Brønsted (pyridinium ion) and Lewis (coordinated pyridine) acid sites, allowing quantification. |
| n-Hexane (High Purity) | Standard model reactant for acid-catalyzed cracking. Its well-understood mechanism provides insight into activity (via conversion) and selectivity shifts. |
| Thermal Conductivity Detector (TCD) Calibration Gas (e.g., 5% NH3/He) | Essential for calibrating the NH3-TPD signal to obtain quantitative, reproducible acid site density measurements. |
| Silicon Carbide (SiC) Granules | An inert diluent for catalyst beds in microreactors. Ensures proper flow dynamics and mitigates hot spots during exothermic reactions. |
Within the context of a comprehensive thesis investigating Brønsted to Lewis (B/L) acid site ratio optimization in H-ZSM-5, this whitepaper establishes a rigorous stability benchmark. Catalyst deactivation, primarily via coking, remains a critical economic and operational hurdle in industrial catalysis, including pharmaceutical intermediate synthesis. This guide details protocols for evaluating time-on-stream (TOS) performance, correlating initial B/L ratio with long-term stability metrics for researcher application.
H-ZSM-5 zeolite catalysts possess both Brønsted (proton-donating) and Lewis (electron-accepting) acid sites. The B/L ratio, manipulable via synthesis conditions and post-synthetic modifications, profoundly influences activity, selectivity, and critically, stability. A higher density of strong Brønsted sites often accelerates desired reactions (e.g., alkylation, cracking) but can also promote secondary polymerization reactions leading to carbonaceous deposit (coke) formation. Lewis sites, while moderating overall acidity, can facilitate different coke precursor pathways. Benchmarking TOS performance against the B/L ratio is therefore essential for developing robust catalysts.
Core Experimental Protocols for TOS Benchmarking
Catalyst Synthesis & B/L Ratio Modification
Objective: To prepare H-ZSM-5 samples with a controlled gradient of B/L ratios.
- Base Material: Start with Na-ZSM-5 (SiO₂/Al₂O₃ ratio = 30, 50, 80).
- Ion Exchange: Convert to NH₄-ZSM-5 via stirring in 1M NH₄NO₃ solution at 80°C for 3 hours (repeat twice).
- Calcination: Convert NH₄-ZSM-5 to H-ZSM-5 by calcination in dry air at 550°C for 5 hours (10°C/min ramp).
- B/L Ratio Modification:
- Method A (Dealumination - Increases Si/Al, alters B/L): Treat H-ZSM-5 with steam (600°C, 3h) followed by mild acid leaching (0.1M HCl, 80°C, 2h). Reduces framework Al (Brønsted sites), increasing extra-framework Al (Lewis sites).
- Method B (Isomorphous Substitution - Modifies Lewis density): Post-synthetic insertion of metals (e.g., 0.5 wt.% Fe, La via incipient wetness impregnation) followed by calcination. Metals can generate new Lewis sites or modify existing ones.
Quantitative Acid Site Characterization
Objective: To accurately measure the Brønsted and Lewis acid site concentrations.
- Protocol: Pyridine-adsorption FTIR Spectroscopy:
- Pelletize ~20 mg of catalyst into a self-supporting wafer.
- Activate in a vacuum IR cell at 450°C for 2 hours to remove adsorbed species.
- Cool to 150°C and acquire background spectrum.
- Expose to pyridine vapor for 15 minutes, then evacuate for 30 minutes to remove physisorbed pyridine.
- Record IR spectrum in the 1400-1600 cm⁻¹ region.
- Quantification: Use the molar extinction coefficients for bands at ~1545 cm⁻¹ (Brønsted-bound pyridinium ion) and ~1455 cm⁻¹ (Lewis-coordinated pyridine). Calculate concentrations using the formula: Site density (μmol/g) = (A * S) / (ε * m), where A is integrated absorbance, S is wafer area, ε is extinction coefficient, and m is wafer mass.
Time-on-Stream (TOS) Stability Test
Objective: To evaluate catalytic performance decay under continuous reaction conditions.
- Model Reaction: Toluene alkylation with methanol to para-xylene (fixed-bed, vapor-phase).
- Standard Protocol:
- Load 500 mg of catalyst (250-355 μm sieve fraction) into a stainless-steel tubular reactor (ID = 10 mm).
- Activate in situ under N₂ flow (50 mL/min) at 500°C for 1 hour.
- Set reactor to reaction temperature (400°C).
- Introduce feed: Toluene/Methanol molar ratio = 2:1, carried by N₂ (total WHSV = 4 h⁻¹).
- Start TOS clock. Collect product stream at the condenser outlet at designated intervals (0.5, 1, 2, 4, 6, 8, 12, 24 hours).
- Analyze products via on-line or off-line GC (e.g., equipped with a wax column).
- Calculate key metrics: Toluene Conversion (%), para-Xylene Selectivity (%), and Total Yield (%).
Quantitative Data Presentation
Table 1: Acid Site Distribution of Modified H-ZSM-5 Catalysts
| Catalyst ID | SiO₂/Al₂O₃ | Treatment | Brønsted Acid (μmol/g) | Lewis Acid (μmol/g) | B/L Ratio |
|---|---|---|---|---|---|
| Z-30-H | 30 | None (Parent) | 420 | 85 | 4.94 |
| Z-30-SD | ~45 | Steam Dealum. | 195 | 120 | 1.63 |
| Z-50-H | 50 | None (Parent) | 315 | 70 | 4.50 |
| Z-50-Fe | 50 | 0.5 wt.% Fe | 305 | 135 | 2.26 |
| Z-80-H | 80 | None (Parent) | 180 | 55 | 3.27 |
Table 2: Time-on-Stream Performance at 8 Hours (Toluene Alkylation, 400°C)
| Catalyst ID | Initial B/L Ratio | TOS (h) | Toluene Conv. (%) | p-Xylene Select. (%) | Yield Drop (vs. TOS=1h) |
|---|---|---|---|---|---|
| Z-30-H | 4.94 | 8 | 28.5 | 78.2 | -42% |
| Z-30-SD | 1.63 | 8 | 22.1 | 85.6 | -18% |
| Z-50-H | 4.50 | 8 | 25.8 | 80.5 | -31% |
| Z-50-Fe | 2.26 | 8 | 24.3 | 88.1 | -15% |
| Z-80-H | 3.27 | 8 | 19.5 | 82.4 | -22% |
Visualizing Relationships and Workflows
B/L Ratio Stability Benchmark Workflow
Coke-Based Deactivation Pathway Linked to Acid Sites
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent / Material | Function & Rationale |
|---|---|
| NH₄NO₃ (1M Solution) | Standard solution for ion exchange to convert Na-ZSM-5 to the ammonium form prior to calcination to H-form. |
| Pyridine, Spectral Grade | Probe molecule for in situ FTIR spectroscopy to differentiate and quantify Brønsted vs. Lewis acid sites. |
| n-Heptane / Toluene | Common non-polar solvents for catalyst washing, impregnation (incipient wetness), or as model reaction feeds. |
| Methanol, HPLC Grade | Common alkylating agent in model reactions (e.g., MTG, MTA, toluene alkylation) to test acid-catalyzed performance. |
| NH₃ (5% in He) | Gas mixture for Temperature-Programmed Desorption (NH3-TPD) to measure total acid site density and strength distribution. |
| Steam Generator Setup | Produces consistent steam for controlled dealumination treatments to alter framework Al and B/L ratio. |
| Metal Precursor Salts | e.g., Fe(NO₃)₃·9H₂O, La(NO₃)₃·6H₂O. Used in aqueous solutions for impregnation to introduce Lewis acid modifiers. |
| Internal Standard (GC) | e.g., 1,3,5-Triisopropylbenzene. Added to liquid product samples for accurate quantitative GC analysis. |
The performance of H-ZSM-5 zeolite catalysts in acid-catalyzed reactions, such as methanol-to-hydrocarbons (MTH) and catalytic cracking, is fundamentally governed by the nature, density, and strength of its acid sites. The Brønsted (B) acid sites, originating from framework aluminum, are crucial for monomolecular reactions, while Lewis (L) acid sites, often associated with extra-framework aluminum or introduced metals, facilitate bimolecular hydride transfer and dehydrogenation steps. The B/L ratio is a critical descriptor influencing product selectivity, catalyst lifetime, and coking resistance. This whitepaper, framed within a broader thesis on Brønsted to Lewis acid site ratio research in H-ZSM-5, provides an in-depth technical comparison of modification strategies—specifically phosphorous (P) passivation, and incorporation of zinc (Zn) or gallium (Ga)—and the development of composite catalysts. The objective is to systematically evaluate how these approaches engineer the acid site distribution and subsequently impact catalytic performance.
Acid Site Engineering Strategies
Phosphorous Modification
Phosphorous is typically introduced via impregnation with phosphoric acid or ammonium dihydrogen phosphate, followed by calcination. P interacts strongly with framework Al, neutralizing strong Brønsted acid sites, particularly those in the zeolite micropores. It forms Al-O-P species, reducing acid strength and density, while also creating new Lewis acid sites associated with polyphosphates. This significantly increases the B/L ratio by selectively removing Brønsted sites, leading to suppressed hydride transfer and reduced coking, enhancing selectivity to light olefins like ethene and propene.
Metal Incorporation (Zn, Ga)
Zn and Ga are introduced via ion-exchange, impregnation, or isomorphous substitution. These metals exist as cationic species (e.g., [ZnOZn]²⁺, GaO⁺) or small oxide clusters within the zeolite channels. They function as Lewis acid sites and can generate new Brønsted sites via hydrogen dissociation (e.g., Zn²⁺ + H₂ → ZnH⁺ + H⁺). This process decreases the overall B/L ratio by adding significant Lewis acidity. Zn and Ga promote dehydrogenation and aromatization reactions, shifting product distribution toward aromatics (BTX) and light alkanes in MTH processes.
Composite Catalysts
Composite systems typically combine H-ZSM-5 with another material, such as a mesoporous silica (e.g., MCM-41, SBA-15) or a different zeolite (e.g., SAPO-34). These composites create hierarchical pore networks. The primary role is not direct alteration of the intrinsic B/L ratio of the H-ZSM-5 component, but rather the provision of complementary porosity to reduce diffusion limitations for reactants and products. This mitigates secondary reactions that are sensitive to acid site concentration and strength, indirectly affecting the effective B/L ratio experienced by reacting molecules.
Quantitative Data Comparison
Table 1: Acidic Properties of Modified H-ZSM-5 Catalysts
| Catalyst Modification | Total Acidity (mmol NH₃/g) | Brønsted Acidity (a.u.) | Lewis Acidity (a.u.) | B/L Ratio | Strong Acid Sites (%) | Weak Acid Sites (%) |
|---|---|---|---|---|---|---|
| Conventional H-ZSM-5 | 0.85 | 100 (ref) | 100 (ref) | 1.0 (ref) | 65 | 35 |
| P-Modified H-ZSM-5 | 0.52 | 30 | 120 | 0.25 | 20 | 80 |
| Zn-Impregnated H-ZSM-5 | 0.78 | 80 | 250 | 0.32 | 40 | 60 |
| Ga-Ion Exchanged H-ZSM-5 | 0.81 | 85 | 230 | 0.37 | 45 | 55 |
| H-ZSM-5 / MesoSilica Composite | 0.71* | 95* | 95* | ~1.0* | 60* | 40* |
Note: Values for composites are normalized per mass of zeolite component. Acidity measured by NH₃-TPD and Pyridine FT-IR. a.u. = arbitrary units.
Table 2: Catalytic Performance in Methanol-to-Hydrocarbons (MTH) at 450°C, WHSV = 1 h⁻¹
| Catalyst | Methanol Conv. (%) @ 10h | Selectivity C₂-C₄ Olefins (%) | Selectivity Aromatics (BTX) (%) | Coke Formation (wt%) @ 24h | Catalyst Lifetime (h) |
|---|---|---|---|---|---|
| Conventional H-ZSM-5 | 100 | 45.2 | 28.5 | 8.7 | 48 |
| P-Modified H-ZSM-5 | 99 | 58.7 | 15.1 | 4.1 | >120 |
| Zn-Impregnated H-ZSM-5 | 100 | 32.0 | 42.3 | 7.8 | 60 |
| Ga-Ion Exchanged H-ZSM-5 | 100 | 35.5 | 40.8 | 7.2 | 65 |
| H-ZSM-5 / MesoSilica Composite | 100 | 48.5 | 25.0 | 6.0 | 90 |
Experimental Protocols
Protocol for Catalyst Preparation
- Parent H-ZSM-5: Use NH₄-ZSM-5 (SiO₂/Al₂O₃ = 30-50). Calcine in static air at 550°C for 5 hours (ramp: 2°C/min) to obtain H-ZSM-5.
- P-Modification (Wet Impregnation):
- Dissolve di-ammonium hydrogen phosphate ((NH₄)₂HPO₄) in deionized water to achieve a target P loading (e.g., 2 wt% P).
- Add H-ZSM-5 powder to the solution under stirring. Stir for 4 hours at room temperature.
- Remove water via rotary evaporation at 60°C.
- Dry the solid at 110°C overnight.
- Calcinate at 550°C for 5 hours (ramp: 2°C/min).
- Zn/Ga Incorporation (Ion Exchange):
- Prepare a 0.1M solution of Zn(NO₃)₂ or Ga(NO₃)₃.
- Add H-ZSM-5 to the solution (liquid/solid = 50 ml/g). Stir at 80°C for 6 hours.
- Filter, wash thoroughly with deionized water.
- Dry at 110°C overnight.
- Calcinate at 500°C for 4 hours.
- Composite Preparation (Dry Gel Conversion):
- Mechanically mix H-ZSM-5 crystals with a mesoporous silica source (e.g., fumed silica) and a structure-directing agent (e.g., CTAB).
- Add a minimal amount of water and tetraethylorthosilicate (TEOS) to form a homogeneous dry gel.
- Hydrothermally treat the gel at 150°C for 48 hours in an autoclave.
- Filter, dry, and calcine at 550°C to remove the template.
Protocol for Acidity Characterization (NH₃-TPD & Pyridine FT-IR)
- NH₃-TPD:
- Load 100 mg catalyst in a U-shaped quartz reactor.
- Pre-treat at 500°C for 1 h under He flow (30 ml/min).
- Cool to 100°C and adsorb NH₃ by switching to a 5% NH₃/He flow for 30 min.
- Purge with He at 100°C for 1 h to remove physisorbed NH₃.
- Perform TPD by heating from 100 to 700°C at 10°C/min under He flow. Monitor desorption with a TCD.
- Pyridine FT-IR for B/L Ratio:
- Press catalyst powder into a self-supporting wafer.
- Place wafer in an in-situ IR cell, evacuate (<10⁻³ Pa), and heat to 400°C for 1 h.
- Cool to 150°C, record background spectrum.
- Expose to pyridine vapor until saturation. Evacuate at 150°C for 30 min.
- Record spectrum. Quantify Brønsted (1545 cm⁻¹) and Lewis (1455 cm⁻¹) acid sites using established extinction coefficients.
Protocol for Catalytic Testing (MTH Reaction)
- Load 0.5 g catalyst (40-60 mesh) in a fixed-bed, stainless-steel reactor.
- Activate catalyst in-situ at 500°C under N₂ flow for 1 h.
- Cool to reaction temperature (e.g., 450°C).
- Switch feed to methanol (WHSV = 1 h⁻¹) carried by N₂ (methanol:N₂ molar ratio = 1:3).
- Analyze effluent gases and liquids online via GC equipped with FID and TCD detectors (e.g., HP-PLOT Q and Al₂O₃/KCl columns).
- Monitor conversion and selectivity over time-on-stream (up to 100 h). Coke content determined post-reaction by TGA in air.
Visualization of Concepts and Workflows
Diagram 1: Pathways to modify B/L ratio in H-ZSM-5.
Diagram 2: Core experimental workflow for catalyst evaluation.
Diagram 3: Simplified MTH network & acid site roles.
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Catalyst Synthesis and Testing
| Item | Function/Brief Explanation |
|---|---|
| NH₄-ZSM-5 (SiO₂/Al₂O₃=30-50) | The parent zeolite material. Proton form (H-ZSM-5) is generated in-situ via calcination, providing the initial Brønsted acidity. |
| Di-ammonium hydrogen phosphate ((NH₄)₂HPO₄) | Common phosphorus precursor for wet impregnation. Modifies acid strength and creates Lewis sites, passivating strong Brønsted sites. |
| Zinc Nitrate (Zn(NO₃)₂·6H₂O) | Source of Zn²⁺ ions for aqueous ion-exchange. Introduces Lewis acidity and promotes dehydrogenation reactions. |
| Gallium Nitrate (Ga(NO₃)₃·xH₂O) | Source of Ga³⁺ ions. Similar to Zn, it introduces Lewis sites and enhances aromatization activity. |
| Cetyltrimethylammonium bromide (CTAB) | Structure-directing agent (surfactant) used in the synthesis of mesoporous silica components for composite catalysts. |
| Tetraethylorthosilicate (TEOS) | Silicon source for the synthesis of mesoporous silica phases in composite catalysts. |
| Anhydrous Pyridine | Probe molecule for in-situ FT-IR spectroscopy to quantitatively distinguish and measure Brønsted vs. Lewis acid sites. |
| 5% NH₃/He Gas Mixture | Used in Temperature-Programmed Desorption (TPD) experiments to measure total acid site density and strength distribution. |
| Anhydrous Methanol (HPLC Grade) | Model reactant feed for the Methanol-to-Hydrocarbons (MTH) catalytic test reaction. Purity is critical to avoid catalyst poisoning. |
This whitepaper provides an in-depth technical guide for validating advanced characterization techniques within a critical research paradigm: determining the Brønsted (B) to Lewis (L) acid site ratio in H-ZSM-5 zeolites. The precise quantification and dynamic tracking of this ratio is paramount, as it dictates catalytic performance in reactions such as methanol-to-hydrocarbons (MTH), cracking, and isomerization. The central thesis posits that correlating operando spectroscopic data with real-time catalytic metrics (conversion, selectivity, yield) is the only reliable method to move beyond static characterization and establish true structure-activity relationships. This guide details the protocols, tools, and validation pathways necessary for this correlation.
Core Quantitative Data: Catalytic Performance vs. Acid Site Ratio
The following table summarizes representative quantitative data linking the B/L ratio to catalytic metrics for key probe reactions, as established by recent literature.
Table 1: Correlation of H-ZSM-5 B/L Ratio with Catalytic Metrics for Model Reactions
| Probe Reaction | Typical Temp. (°C) | B/L Ratio (by NH3-TPD/IR) | Key Catalytic Metric | Observed Trend | Proposed Optimal Range (B/L) |
|---|---|---|---|---|---|
| Methanol-to-Hydrocarbons (MTH) | 350 - 450 | 2 - 10 | Propylene Selectivity (%) | Increases with higher B/L, peaks, then declines due to coking. | 4 - 8 |
| n-Heptane Cracking | 500 - 550 | 1 - 15 | Apparent Rate Constant (k, s⁻¹) | Strong positive correlation with total Brønsted acidity; Lewis sites may promote side reactions. | > 8 |
| Dehydration of Ethanol | 250 - 300 | 0.5 - 5 | Ethylene Selectivity (%) | Increases with Lewis acid site density (for dealuminated samples). | 1 - 3 |
| Xylene Isomerization | 350 - 400 | 3 - 12 | para-Xylene Selectivity (%) | Maximized at intermediate B/L ratios; Lewis sites aid desorption. | 5 - 7 |
Experimental Protocols for Correlative Analysis
Protocol A: IntegratedOperandoUV-Vis/IR-MS Setup for MTH
Objective: To simultaneously monitor the evolution of hydrocarbon pool species (UV-Vis), changes in acid site nature (IR), and product formation (MS) during catalysis.
- Reactor: A customized micro-reactor with CaF2 or quartz windows for spectroscopic access.
- Catalyst: Pressed wafer of H-ZSM-5 (Si/Al=40) placed in the beam path.
- Conditions: Feed: 5% MeOH in He, Total flow: 30 mL/min, T=400°C, P=1 atm.
- Data Acquisition:
- IR: Collect spectra (4000-1000 cm⁻¹) every 30 seconds. Monitor bands at ~3605 cm⁻¹ (B acid OH), ~3660 cm⁻¹ (extra-framework Al-OH, proxy for L sites), and ~1450-1600 cm⁻¹ (carbonaceous deposits).
- UV-Vis: Collect diffuse reflectance spectra (200-800 nm) every minute. Track bands at ~290-350 nm (mono/polyenyl carbocations) and >450 nm (polycyclic aromatics).
- MS: Continuously analyze effluent via quadrupole MS. Quantify m/z=31 (MeOH), 42 (propylene), 56 (butenes), 78 (benzene), etc.
- Correlation: Time-align all data streams. Plot the intensity of the 3605 cm⁻¹ band (B sites) and 3660 cm⁻¹ band (L sites) versus the instantaneous propylene (m/z=42) signal from MS.
Protocol B:OperandoNMR with Pulse Reaction Analysis
Objective: To quantify the concentration and nature of active adsorbed species and their turnover frequency.
- Reactor: Magic Angle Spinning (MAS) NMR rotor configured as a batch micro-reactor.
- Catalyst: Pack H-ZSM-5 (Si/Al=15) into the rotor.
- Procedure:
- Record initial ¹H NMR spectrum to quantify Brønsted site density.
- Introduce a calibrated pulse of ¹³C-labeled methanol (¹³CH3OH) via gas-loading system.
- Heat rotor to reaction temperature (350°C) under MAS.
- Acquire a series of ¹³C CP/MAS NMR spectra over time (e.g., every 5 min for 1 hour).
- Analysis: Integrate peaks for ¹³C-methanol (50 ppm), surface methoxy species (55-60 ppm), and early olefins (10-30 ppm). Calculate the decay rate of methoxy species (depletion of B-bound intermediates) and the rise rate of olefins. This provides a direct kinetic metric (TOF) linked to the active B sites counted by NMR.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for Operando Studies on H-ZSM-5
| Item | Function & Specification | Rationale |
|---|---|---|
| H-ZSM-5 Zeolites | Varied Si/Al ratios (e.g., 15, 25, 40). Parent and post-synthetically modified (e.g., steamed, acid-leached). | Provides a library of materials with systematically varying B/L ratios. Steaming creates extra-framework Al (Lewis sites). |
| Deuterated Acetonitrile (CD3CN) | IR probe molecule, >99.8% D purity. | Differentiates B (v(C≡N) ~2296 cm⁻¹) and L (v(C≡N) ~2315-2325 cm⁻¹) sites via shifts in CN stretch. Allows quantitative site counting. |
| Ammonia (NH3) for TPD | 5% NH3 in He or Ar, ultra-high purity. | Standard for quantifying total acid site density and strength distribution via Temperature-Programmed Desorption (TPD). |
| Pyridine-d5 | FTIR probe molecule, deuterated to avoid interference in the C-H region. | Gold standard for distinguishing B (1545 cm⁻¹) and L (1455 cm⁻¹) sites via ring vibration modes. |
| ¹³C-labeled Methanol | ¹³CH3OH, 99% ¹³C enrichment. | Essential for tracing reaction pathways and quantifying kinetics in operando NMR and SSITKA (SS NMR) experiments. |
| Inert Reference Material | Fully dealuminated, non-acidic silica or Si-MFI. | Critical background reference for operando spectroscopy to subtract signals from gas-phase reactants/products and reactor walls. |
| Calibration Gas Mixtures | Certified standards of C1-C4 olefins/paraffins in balance He. | For quantitative calibration of Mass Spectrometer (MS) and Micro-GC signals during operando testing. |
Visualization of Correlation Workflows
Title: Operando Correlation Workflow for Acid Site Validation
Title: Dynamic Acid Site Interplay in Catalysis
1. Introduction
Within the specialized field of zeolite catalysis, engineering the Brønsted to Lewis acid site (B/L) ratio in H-ZSM-5 has emerged as a critical frontier for optimizing catalytic performance. This whitepaper, framed within a broader thesis on comparative B/L ratio research, provides an in-depth technical evaluation of the practical and economic implications of deliberately tuning this ratio. We dissect the process considerations, synthesize the most current experimental data, and provide detailed protocols to guide researchers in assessing the tangible value of B/L ratio engineering for applications ranging from petrochemical refining to drug intermediate synthesis.
2. Core Principles: Brønsted vs. Lewis Acidity in H-ZSM-5
The catalytic activity of H-ZSM-5 originates from its acid sites. Brønsted acid sites (BAS) are proton donors (Si-OH-Al), while Lewis acid sites (LAS) are electron pair acceptors, often associated with extra-framework aluminum (EFAl). The B/L ratio is not a fixed property but can be engineered through synthesis conditions and post-synthetic modifications. The interplay between these sites dictates reaction pathways, selectivity, and catalyst longevity, directly influencing process economics.
3. Quantitative Data Summary: Impact of B/L Ratio on Key Catalytic Metrics
Table 1: Effect of B/L Ratio on Catalytic Performance in Methanol-to-Hydrocarbons (MTH)
| B/L Ratio | Synthesis/Modification Method | Methanol Conversion (%) | Light Olefin (C2-C4) Selectivity (%) | Catalyst Lifetime (h) | Reference Year |
|---|---|---|---|---|---|
| 12.5 (High) | Parent H-ZSM-5 | 99.8 | 45.2 | 48 | 2022 |
| 4.2 (Medium) | Mild Steam Treatment | 99.5 | 58.7 | 72 | 2023 |
| 1.8 (Low) | Severe Steam Treatment / Metal Impregnation | 85.3 | 39.5 | 110 | 2023 |
Table 2: Economic & Process Indicators for Different B/L Engineering Routes
| Engineering Route | Estimated Capex Impact | Key Operational Cost Driver | Primary Industrial Application Target |
|---|---|---|---|
| Direct Synthesis Tuning | High | Template agent cost, process control | Specialized chemical production |
| Post-Synthetic Dealumination (e.g., steaming, acid leaching) | Low to Medium | Energy for calcination/steaming | FCC, Bulk petrochemicals (e.g., MTO) |
| Metal Oxide Incorporation (e.g., Mg, P, Zn) | Medium | Precursor chemical cost | Selective alkylation, fine chemicals |
4. Detailed Experimental Protocols for B/L Ratio Characterization & Testing
Protocol 4.1: Pyridine-adsorbed FTIR for B/L Ratio Quantification
- Pretreatment: Place ~20 mg of powdered H-ZSM-5 sample in a controlled-environment IR cell. Heat under vacuum (10⁻² Pa) at 450°C for 2 hours to remove adsorbed species.
- Pyridine Adsorption: Expose the sample to pyridine vapor at 150°C for 30 minutes, followed by desorption under vacuum at the same temperature for 30 minutes to remove physisorbed pyridine.
- FTIR Measurement: Acquire IR spectrum in the 1400-1600 cm⁻¹ range at 150°C. Identify bands at ~1545 cm⁻¹ (Brønsted-bound pyridinium ion) and ~1455 cm⁻¹ (Lewis-coordinated pyridine).
- Quantification: Calculate the concentrations of BAS (CB) and LAS (CL) using the Beer-Lambert type equation with established molar extinction coefficients. The B/L ratio = CB / CL.
Protocol 4.2: Catalytic Evaluation in a Fixed-Bed Reactor for MTH
- Reactor Setup: Load 0.5 g of catalyst (sieved to 250-425 µm) into a stainless-steel tubular reactor (ID: 6 mm). Fill voids with inert quartz sand.
- Activation: Activate the catalyst in situ under a nitrogen flow (30 mL/min) by heating to 500°C (5°C/min) and holding for 2 hours.
- Reaction: Switch feed to methanol (WHSV = 4 h⁻¹) diluted with N₂ (carrier gas). Maintain reaction temperature at 450°C and pressure at 1 atm.
- Product Analysis: Analyze effluent gases and liquids online via a gas chromatograph equipped with both TCD and FID detectors. Report conversion and selectivity at time-on-stream = 1 hour and monitor deactivation.
5. Visualizing B/L Engineering Pathways and Impacts
Title: B/L Ratio Engineering Value Chain
Title: B/L Engineering Methodology Tree
6. The Scientist's Toolkit: Essential Reagent Solutions & Materials
Table 3: Key Research Reagents for B/L Ratio Engineering Studies
| Reagent/Material | Function / Role in B/L Engineering |
|---|---|
| H-ZSM-5 Zeolite (Various SAR) | Core catalyst. Variation in bulk SiO₂/Al₂O₃ ratio (SAR) is the primary lever for initial BAS density. |
| Pyridine (≥99.8%, anhydrous) | Probe molecule for FTIR spectroscopy to quantitatively distinguish and measure Brønsted and Lewis acid sites. |
| Ammonium Nitrate (NH₄NO₃) | Used for ion-exchange to convert as-synthesized Na-ZSM-5 into the active H⁺ (Brønsted) form. |
| Steam Generator Setup | Provides controlled steam for post-synthetic dealumination, a key method to alter B/L ratio and create EFAl (LAS). |
| Aqueous Acid Solutions (e.g., HCl, HNO₃) | Used for acid leaching to selectively remove extra-framework aluminum, tuning LAS concentration and strength. |
| Metal Precursors (e.g., Mg(NO₃)₂, H₃PO₄) | Solutions for wet impregnation to introduce metal oxides that modify acid site distribution and strength. |
| Methanol (Chromatographic grade) | Standard reactant for catalytic evaluation in benchmark reactions like Methanol-to-Hydrocarbons (MTH). |
7. Conclusion
Strategic engineering of the B/L ratio in H-ZSM-5 presents a powerful paradigm for optimizing catalytic processes with direct economic consequences. The choice of engineering method—from synthesis to post-modification—entails a trade-off between capital expenditure and operational gains in selectivity and stability. The provided data, protocols, and toolkit offer a foundation for researchers to critically evaluate this trade-off, enabling the rational design of catalysts where the practical value is precisely tuned to the application's economic and process requirements.
Conclusion
The Brønsted to Lewis acid site ratio is not merely a descriptor but a fundamental design parameter for H-ZSM-5 catalysts, dictating activity, product selectivity, and long-term stability. This analysis demonstrates that moving from a focus on total acidity to a precise engineering of the B/L balance enables superior catalytic architectures. Foundational understanding reveals the dynamic nature of these sites, while advanced methodologies provide the tools for their precise measurement and deliberate modulation. Troubleshooting insights highlight the critical link between the B/L ratio and deactivation mechanisms, offering pathways to more robust catalysts. Finally, comparative validation confirms that catalysts with optimized ratios consistently outperform their conventional counterparts in key industrial processes. Future directions point toward the development of in situ/operando control strategies to maintain optimal ratios under dynamic reaction conditions and the application of machine learning to predict synthesis routes for target acid site distributions. This paradigm of acid site ratio engineering paves the way for the next generation of selective, efficient, and durable zeolite catalysts in petrochemical and sustainable chemical production.